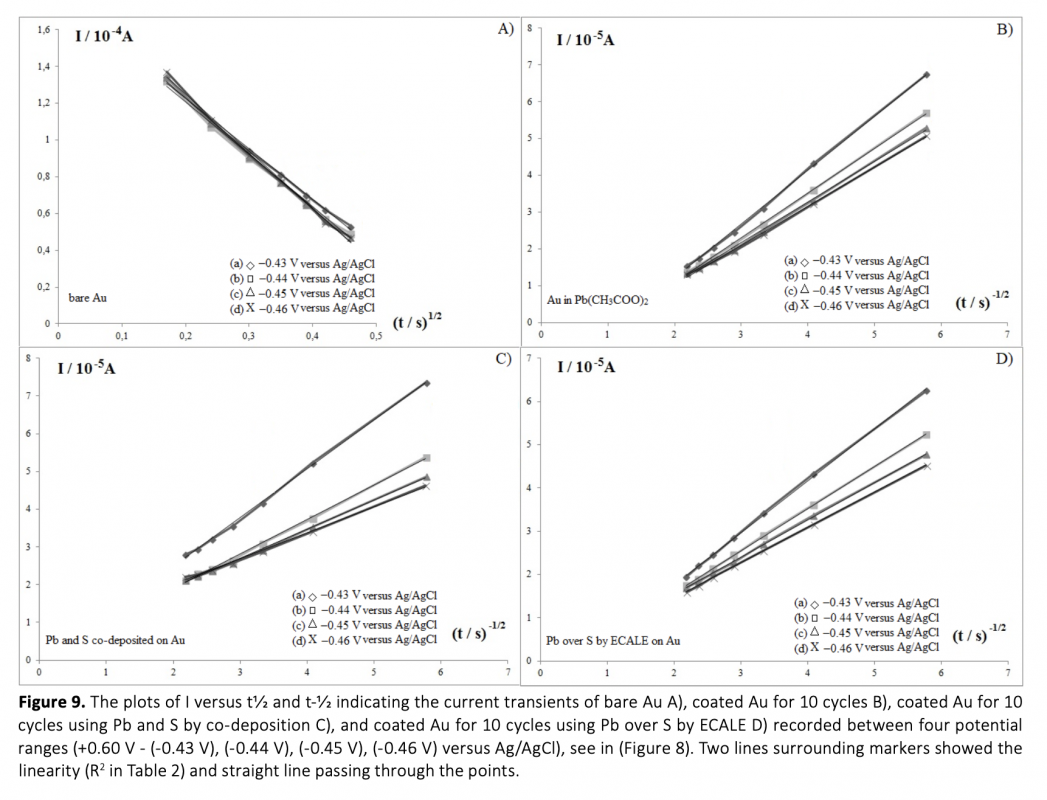
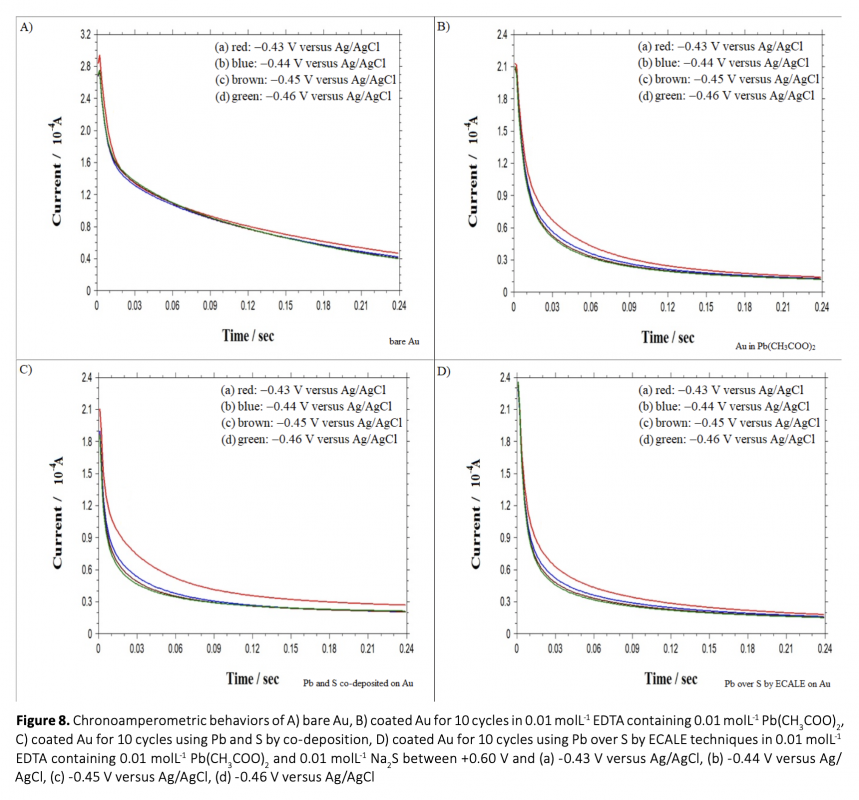
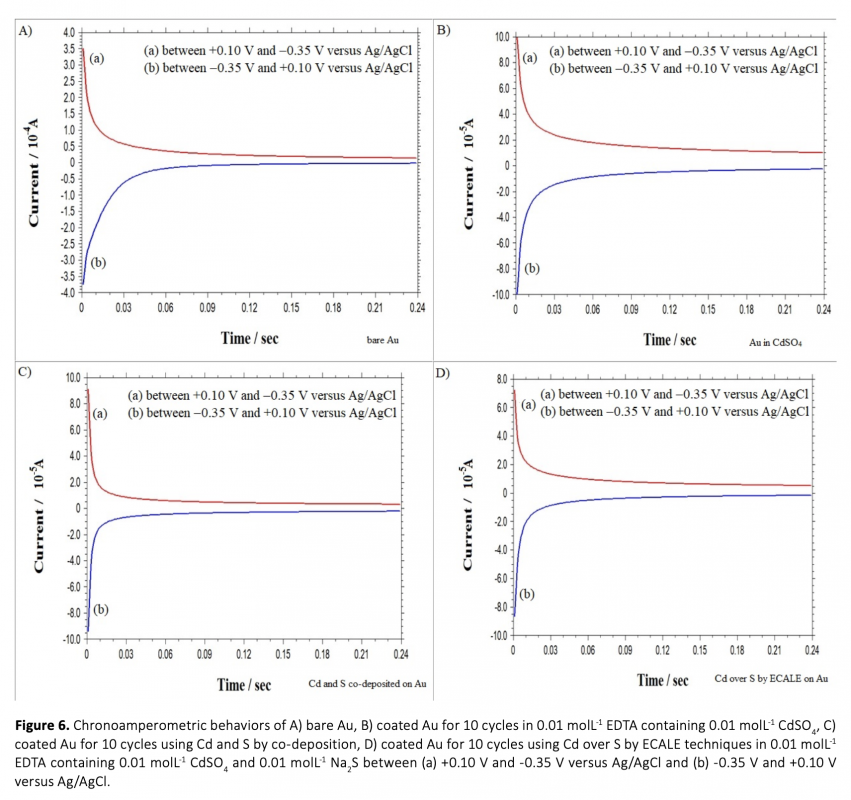
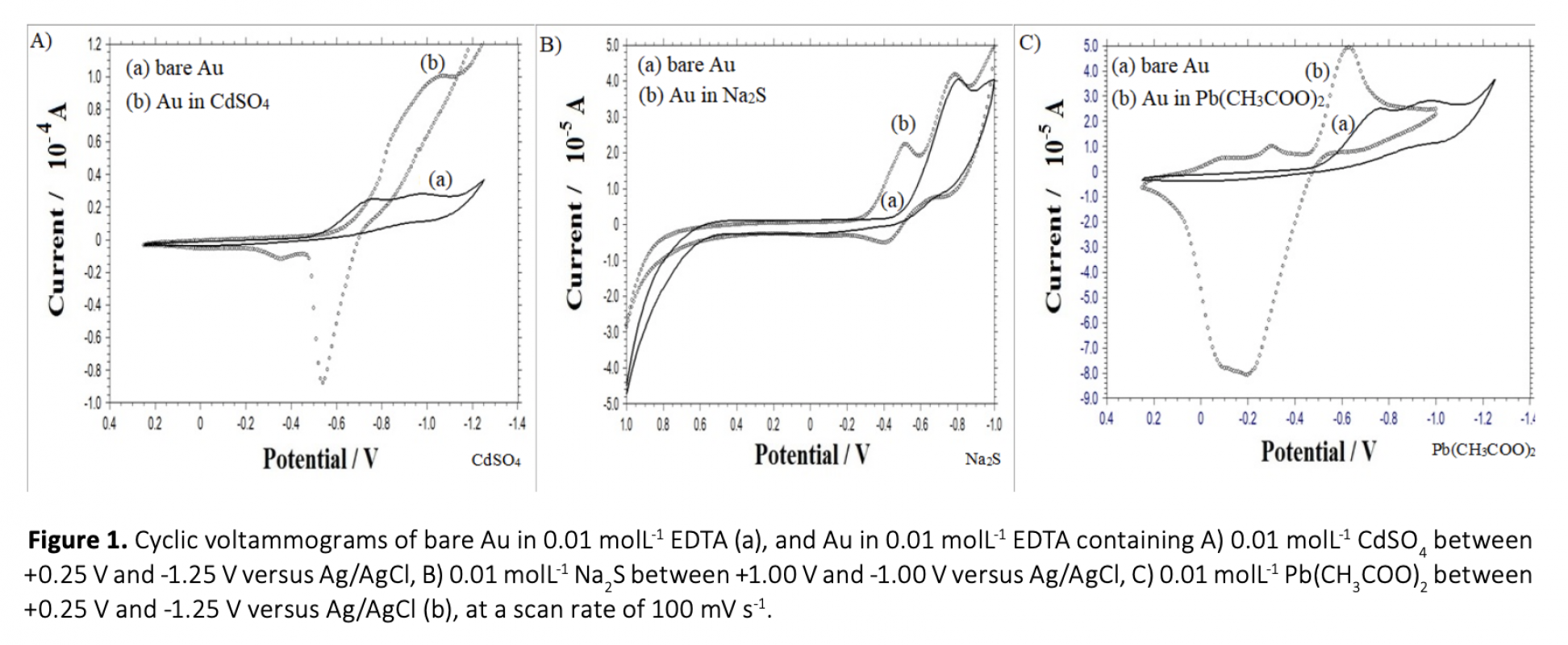
Anew, simple and cost-effective electrochemical route was demontrated in this work. CdS and PbS thin films were grown on polycrystalline gold electrode using co-deposition and ECALE techniques based on accumulation layer by layer. The deposition potentials of cadmium, lead and sulfur were determined separately by cyclic voltammetry. Thin films were created from an electrolyte containing 0.01 molL-1 CdSO4, 0.01 molL-1 Na2S and 0.01 molL-1 Pb(CH3COO)2 in 0.01 molL-1 EDTA (pH = 3.00). The influence of bath temperature at the deposition potential was studied to determine the crystallinity of deposits. From the chronoamperometry results including the transients which were obtained within the under potential region, the overall shape of the experimental depositions was proposed and the growth process was considered.
Bu çalışmada yeni, basit ve uygun maliyetli bir elektrokimyasal rutin gösterildi. CdS ve PbS ince filmler polikristalin altın elektrot üzerinde tabaka tabaka biriktirme temeliyle ko-depozisyon ve ECALE teknikleri kullanılarak büyütüldü. Kadmiyum, kurşun ve kükürdün biriktirme potansiyelleri ayrı ayrı dönüşümlü voltametri ile belirlendi. İnce filmler 0.01 molL-1 EDTA’da (pH = 3.00) 0.01 molL-1 CdSO4, 0.01 molL-1 Na2S ve 0.01 molL-1 Pb(CH3COO)2 içeren elektrolitten oluşturuldular. Biriktirme potansiyelindeki sıcaklık etkisi birikintilerin kristalliğinin belirlenmesi için çalışıldı. Potansiyel altı alanı içerisinde elde edilen geçişleri kapsayan kronoamperometri sonuçlarından, deneysel birikimlerin genel şekilleri önerildi ve büyüme süreci değerlendirildi.
Download Article in PDF (4.0 MB)