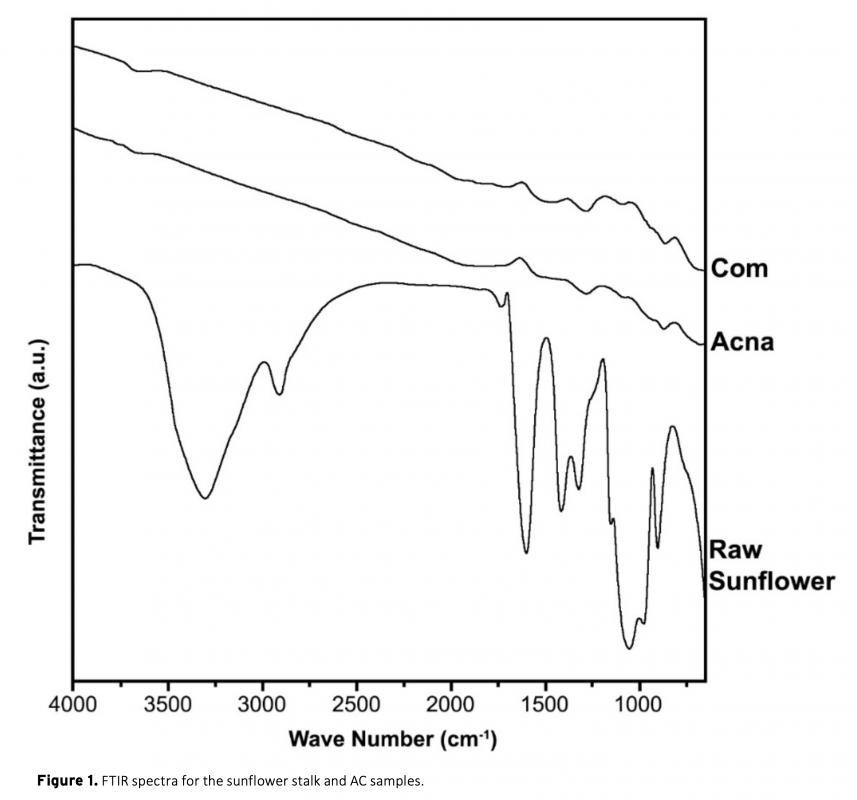
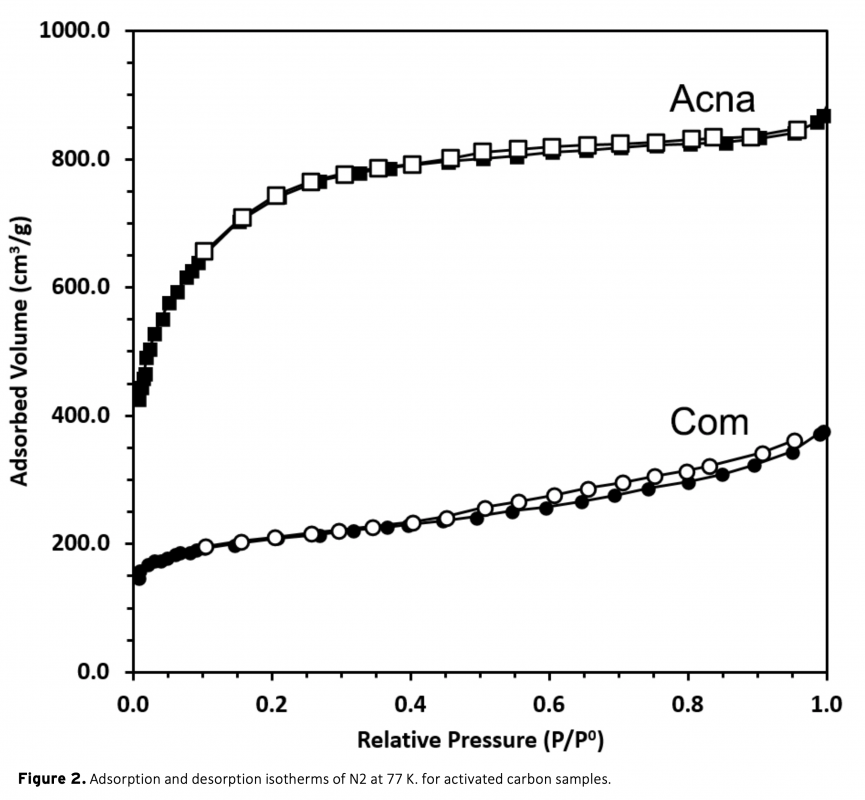
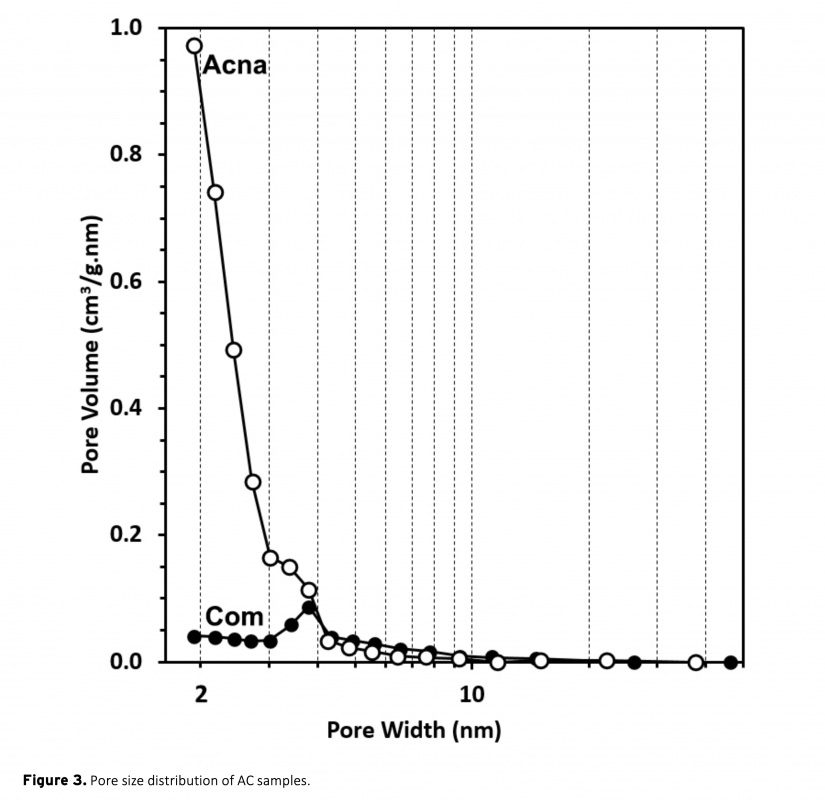
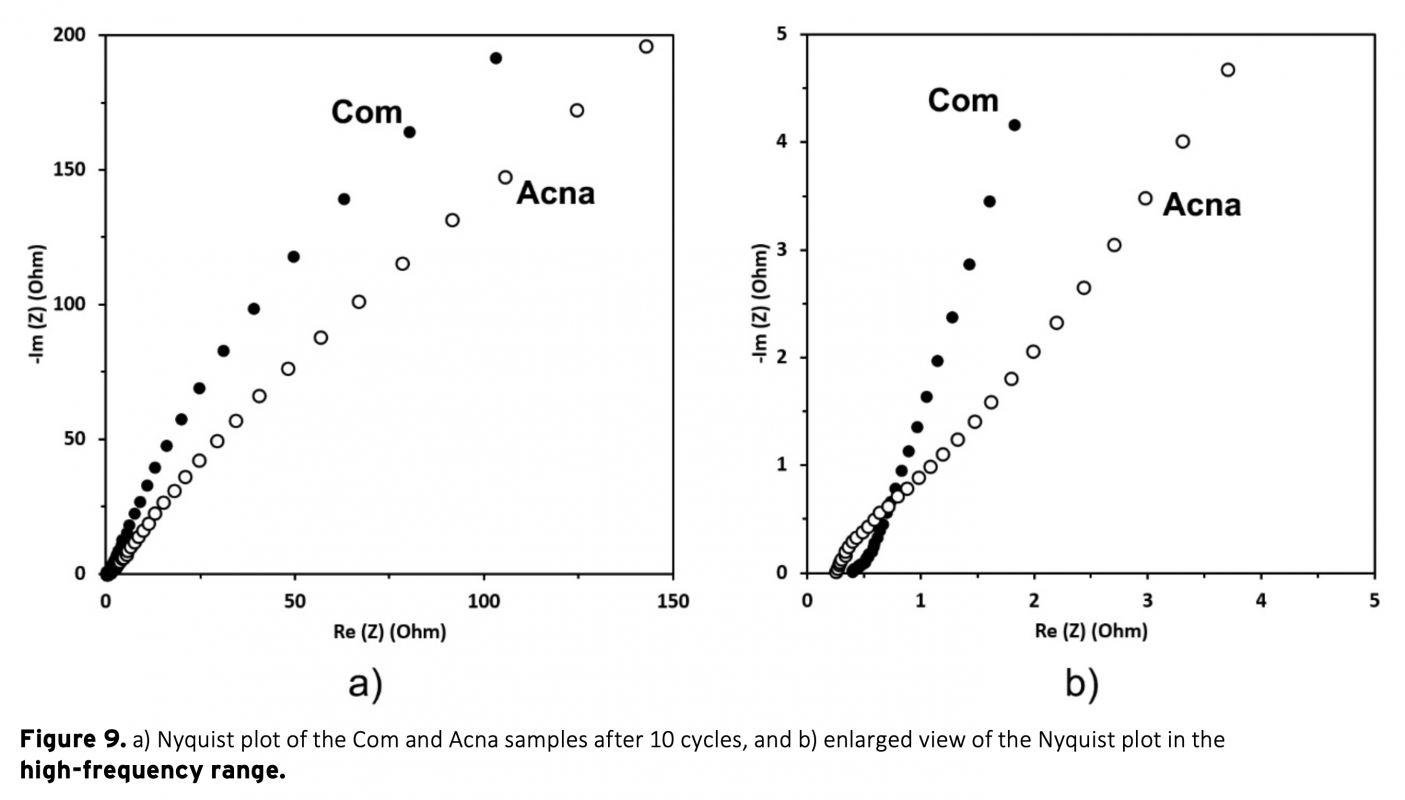
In this study, a combination of physical and chemical activation was used to produce activated carbon from sunflower stalks. The NaOH activated carbon possess a high specific surface area of 2658 m2/g. The micropore fraction and surface area obtained is much higher than a commercial activated carbon. The electrodes from the activated carbons were electrochemically analyzed in a two-electrode supercapacitor cell with 1 M H2SO4 electrolyte. The results show that the high surface area of sunflower activated carbon resulted in significantly high specific capacitance of 207 F/g at 0.05 A/g current density. Moreover, a high energy density of 18.4 Wh/kg was obtained at the power density of 80 W/kg. The results also showed the importance of pore structure on the supercapacitor performance.
Bu çalışmada, ayçiçeği saplarından aktif karbon üretmek için fiziksel ve kimyasal aktivasyon yöntemlerinin bir bileşimi kullanıldı. NaOH ile aktive edilen aktif karbon, 2658 m2/g gibi yüksek bir yüzey alanına sahiptir. Elde edilen mikrogözenek oranı ve yüzey alanı mktarı ticari aktif karbonlardan çok daha yüksektir. Aktif karbonlardan elde edilen elektrotlar, ikili süper kapasitör hücresinde, 1 M H2SO4 elektrolit ile elektrokimyasal olarak analiz edilmiştir. Sonuçlar, ayçiçeğinden elde edilen yüksek yüzey alanı sayesinde, 0.05 A/g akım yoğunluğunda, önemli derecede yüksek bir kapasitans olan 207 F/g elde edildiğini göstermiştir. Buna ek olarak, 80 W/kg güç yoğunluğunda yüksek bir enerji yoğunluğu değeri olan 18.4 Wh/kg elde edilmiştir. Sonuçlar ayrıca gözenek yapısının süper kapasitör performansı için ne kadar önemli olduğunu da göstermiştir.
Download Article in PDF (1.0 MB)