The electrochemical behavior of amlodipine (AML) and telmisartan (TLM) on a glassy carbon electrode were investigated in different aqueous solutions by using cyclic, differential pulse, and square wave voltammetric techniques. AML and TLM exhibited one peak (both of them) to the anodic direction. The oxidation process was found to be irreversible and adsorption controlled. To obtain good sensitivity, the instrumental and accumulation variables were studied using square wave voltammetry (SWV) techniques in 0.5 M H2SO4 solution, which was obtained as the most repeatabile and sensitive media. In this study; sensitive, fast and reliable method development was aimed for simultaneous detection of AML and TLM. This voltammetric method exhibited linear dynamic responses in the concentration range between 1.0×10-7 M and 1.0×10-4 M for AML and 1.0×10-7 M and 1.0×10-5 M for TLM, with detection limits of 0.654 nM and 22.6 nM, respectively. The proposed method is validated and successfully applied for the analysis of AML and TLM tablets.
Amlodipine (AML) ve telmisartan (TLM)’ın elektrokimyasal davranışı; farklı sulu çözeltilerde, camsı karbon elektrotta, dönüşümlü, diferansiyel puls ve kare dalga voltametri (KDV) teknikleri kullanılarak araştırılmıştır. AML ve TLM anodik yönde bir pik (her ikisi de) vermiştir. Oksidasyon prosesi geri dönüşümsüz ve adsorpsiyon kontrollü olarak bulunmuştur. En tekrar edilebilir ve duyarlı ortam olarak 0.5 M H2SO4 çözeltisi bulunmuş ve bu ortamda iyi bir hassasiyet elde edebilmek için enstrümantal ve biriktirme değişkenleri KDV tekniği ile çalışılmııştır. Bu çalışmada, AML ve TLM’ın eş zamanlı tayini için duyarlı, hızlı ve güvenilir yöntemin geliştirilmesi amaçlanmıştır. Bu voltametrik yöntem, 1.0×10-7 M ve 1.0×10-4 M aralığında AML için ve 1.0×10-7 M ve 1.0×10-5 M aralığında TLM için doğrusal dinamik cevaplar vermiş olup gözlenebilme sınırları AML ve TLM için sırasıyla 0.654 nM, 22.6 nM olarak bulunmuştur. Önerilen yöntem, AML ve TLM tabletlerinden analiz çalışmalarına başarıyla uygulanmış olup tamamen doğrulanmıştır.
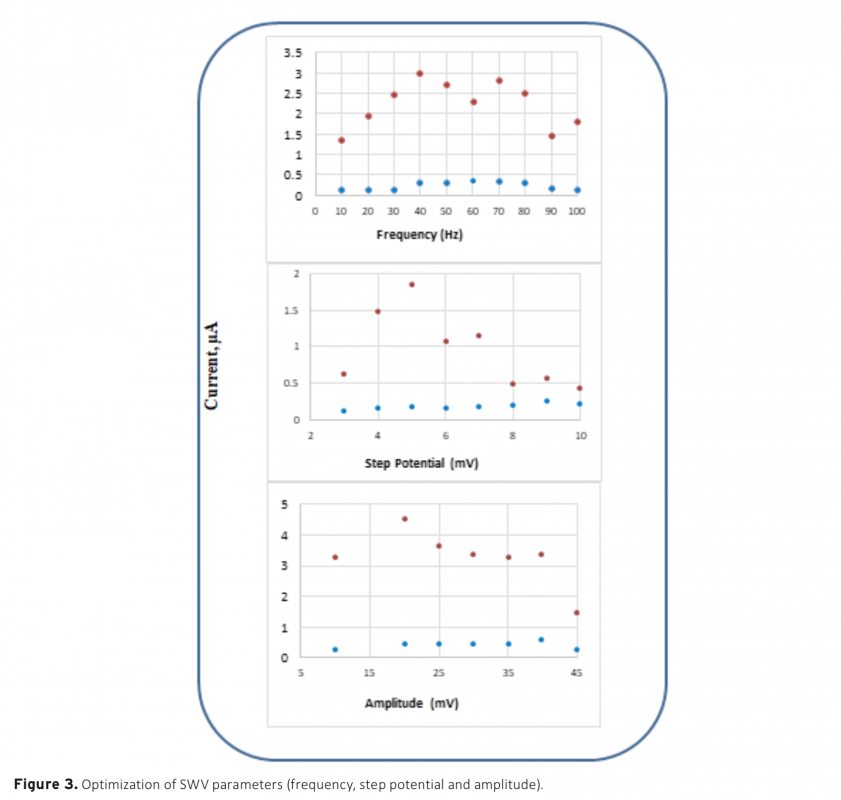
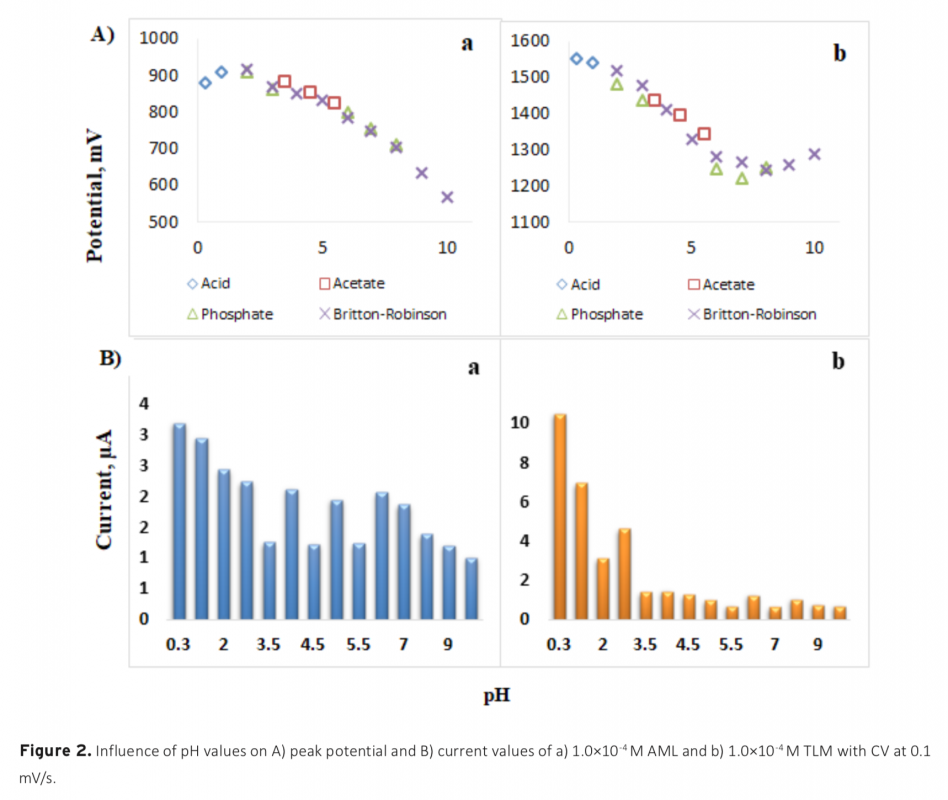
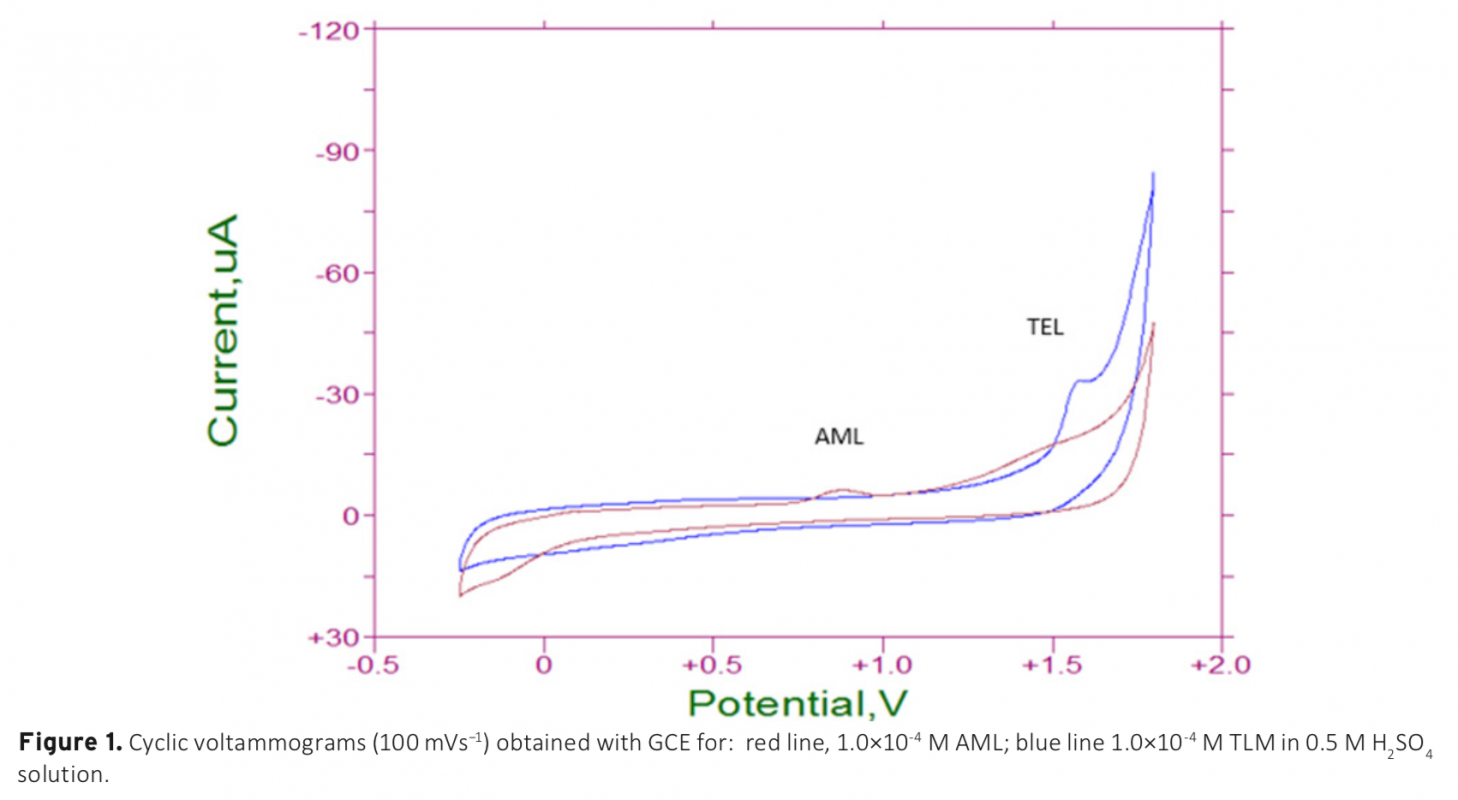
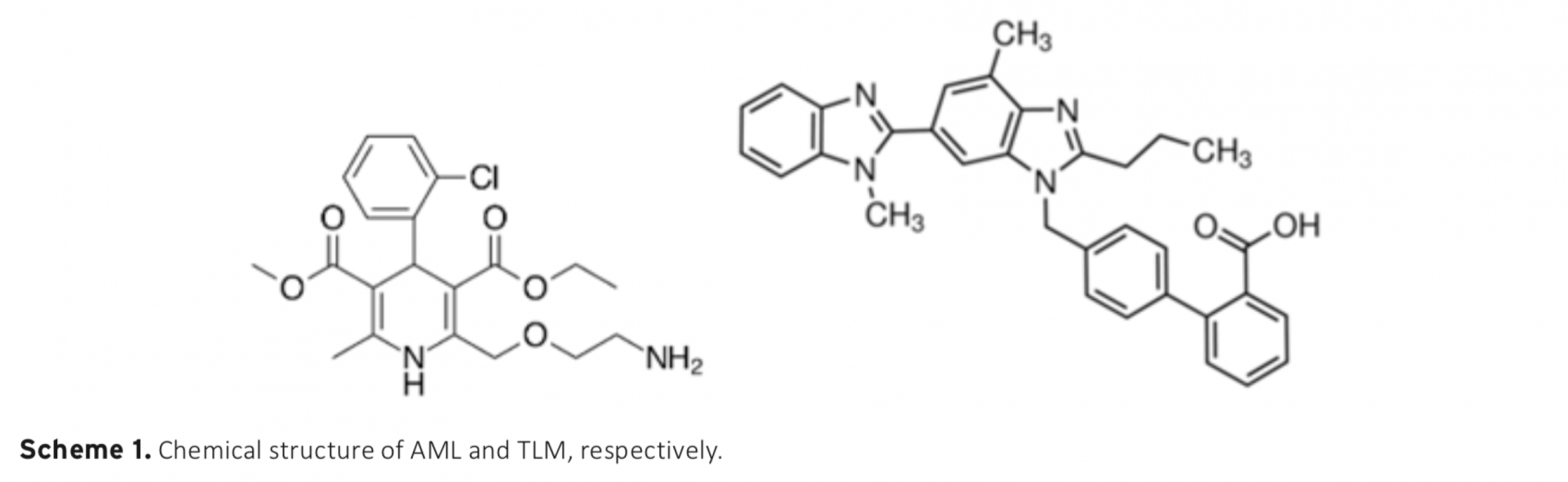
Download Article in PDF (515.0 kB)