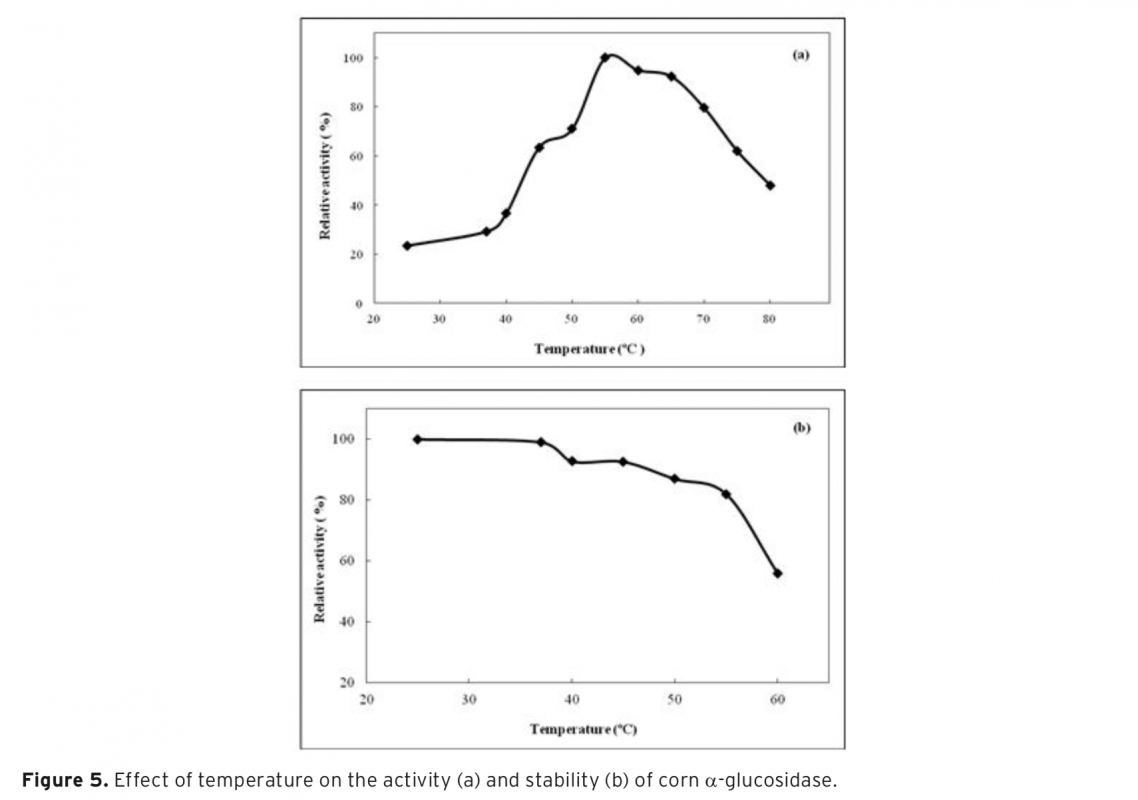
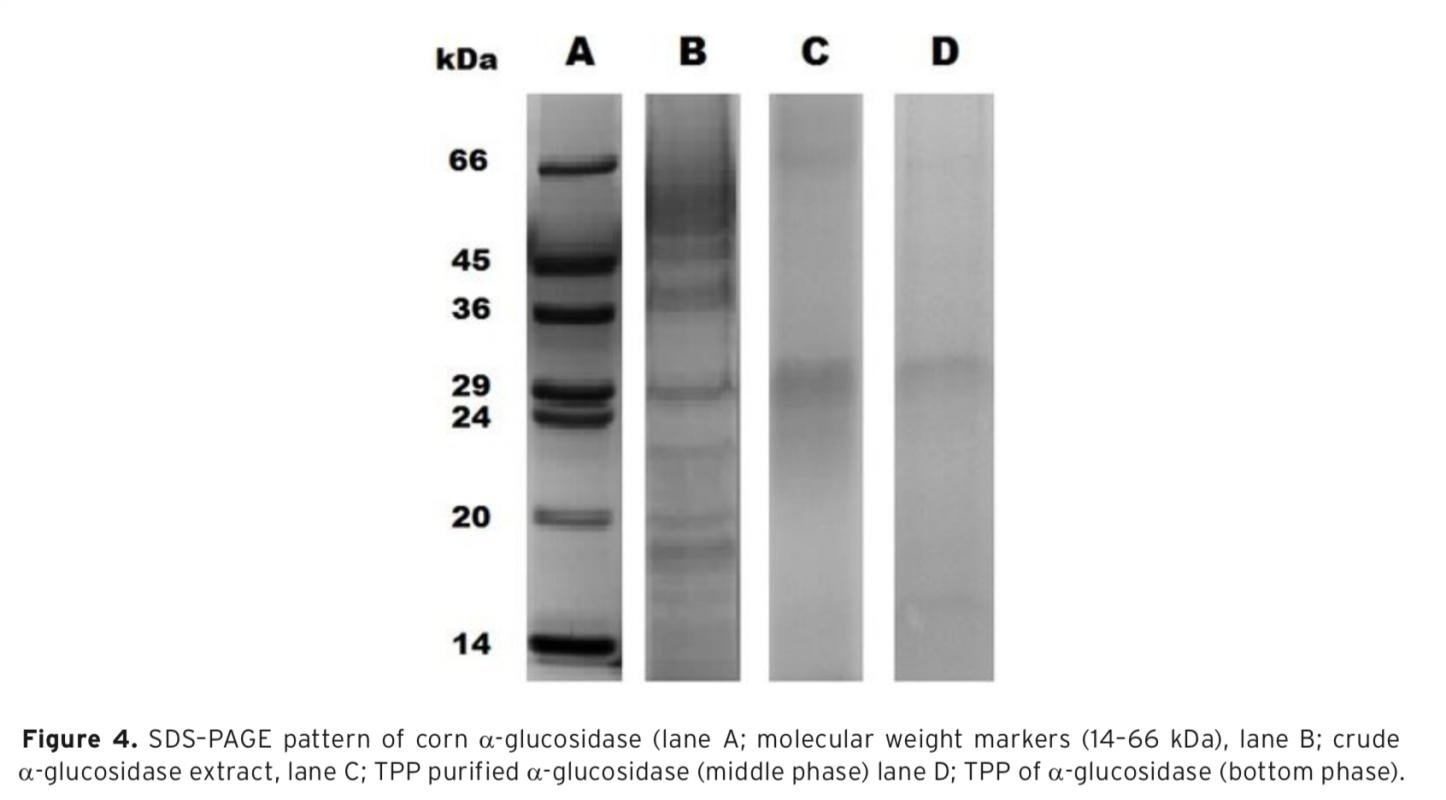
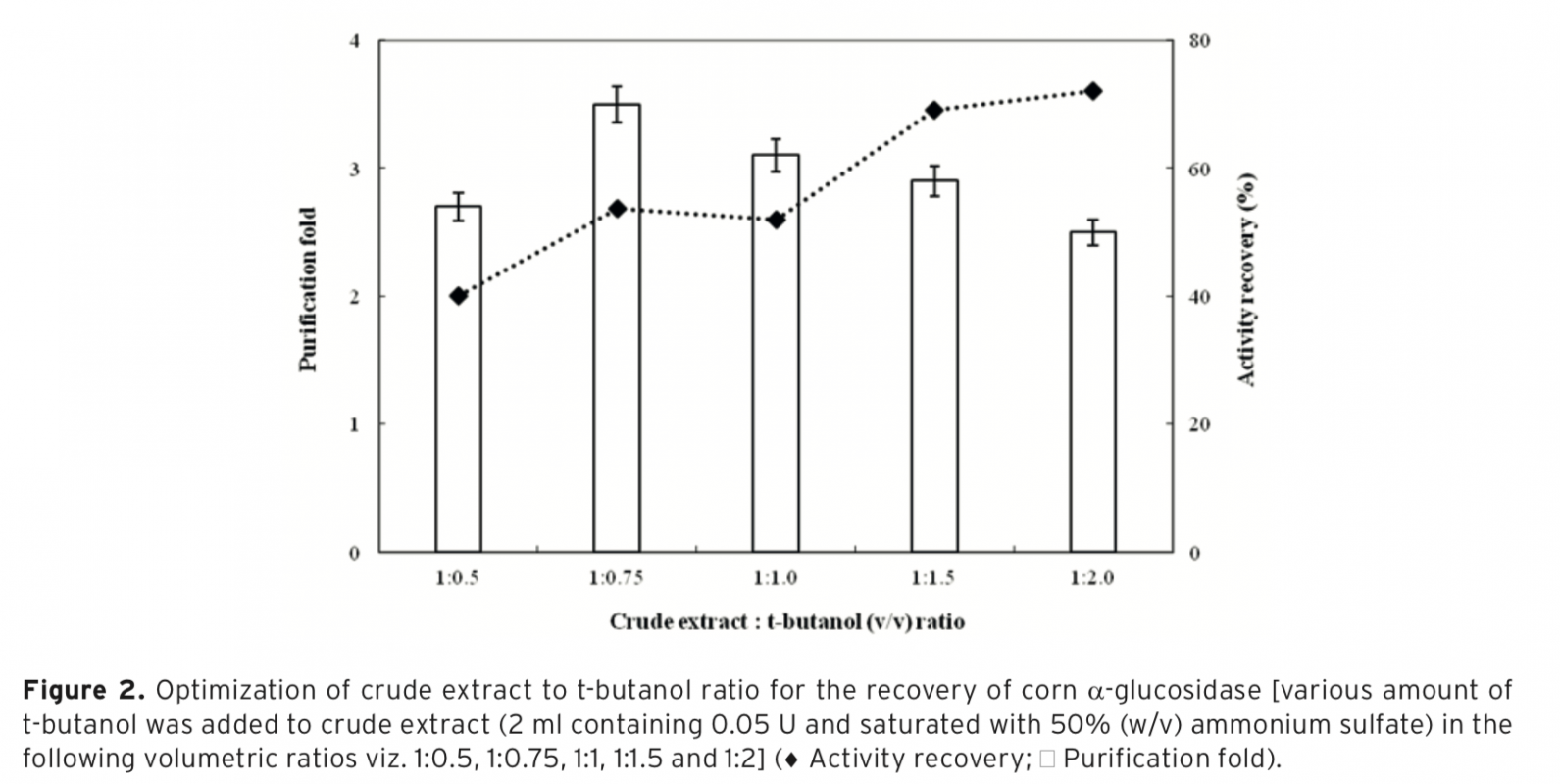
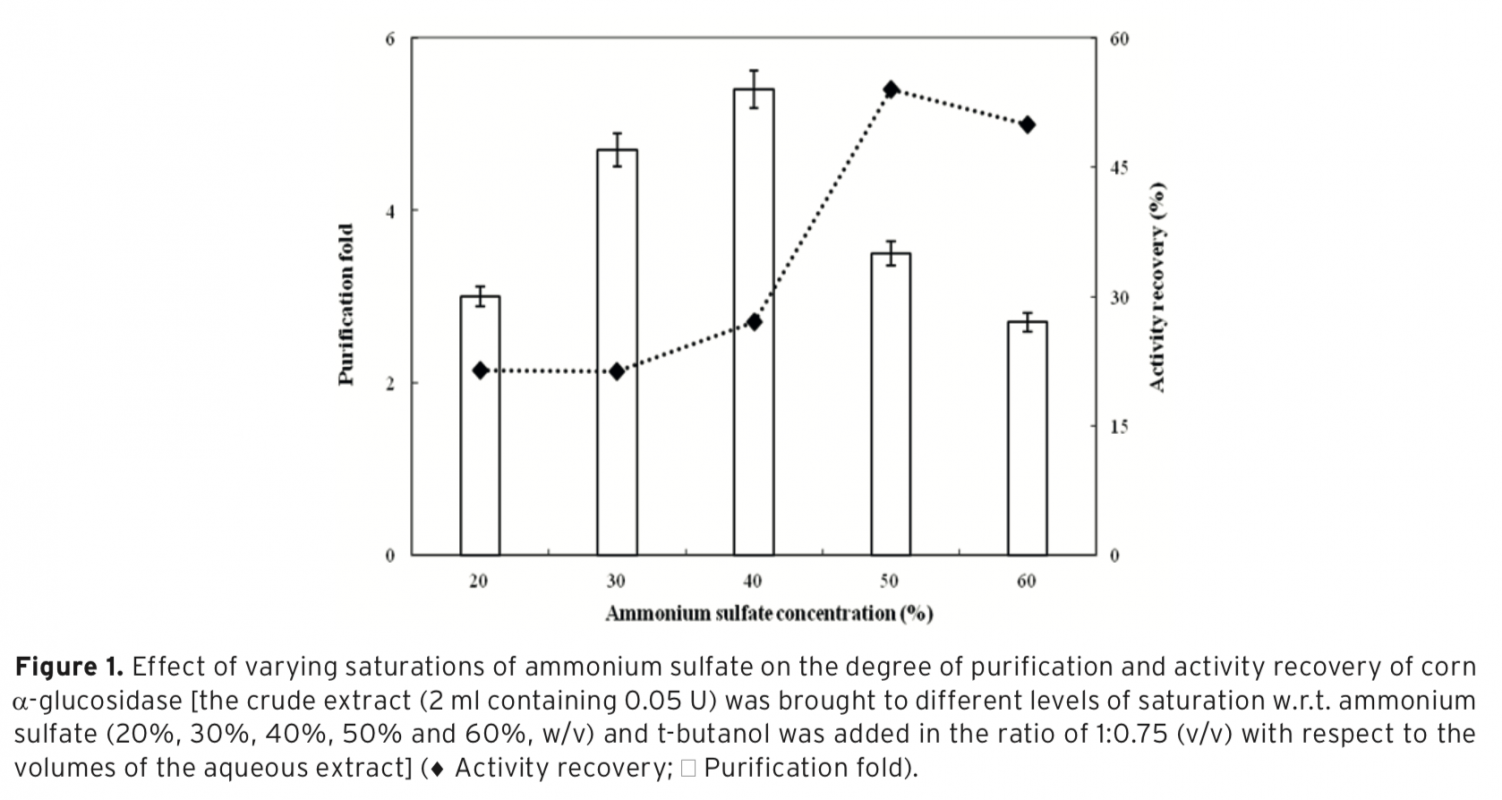
α-Glucosidase was first purified from corn by three-phase partitioning(TPP) and then characterized. Ammonium sulfate and t-butanol were used in order to obtain three phases. Effect of different process parameters such as; ammonium sulfate concentration, enzyme to t-butanol ratio and pH required for efficient purification of the corn α-glucosidase was studied to get highest purification fold and activity recovery. Optimum purification parameters of the TPP system were determined as 50% (w/v) ammonium sulfate saturation with 1.0:0.75 (v/v) ratio of crude extract: t-butanol at pH 4.5. Under optimized conditions α-glucosidase was purified with 3.8 purification fold and 60% activity recovery. The molecular weight of α-glucosidase was determined approximately as 29 kDa by using sodium dodecyl sulfate–polyacrylamide gel electrophoresis(SDS–PAGE). Characterization studies showed that, optimum pH and temperature of α-glucosidase were pH 4.5 and 55°C, respectively. The purified enzyme was found to be very stable at a temperature range of 25-60°C and a pH range of 3.5-5.5. Kinetic constants (KM and Vmax) were calculated from Lineweaver-Burk plot as 0.64 mM and 0.45 U, respectively. With 10 mM of CaCl2 concentration α-glucosidase activity was significantly increased to 130%. TPP is a useful strategy to concentrate and purify α-glucosidase for its applications.
α-Glukozidaz önce mısırdan üçlü-faz ayırma (TPP) tekniği ile saflaştırıldı ve ardından karakterize edildi. Üç faz elde etmek için amonyum sülfat ve t-butanol kullanıldı. Mısır α-glukozidazının yüksek saflaştırma katı ve aktivite geri kazanımı ile etkin bir şekilde saflaştırılması için çeşitli proses parametrelerinin örneğin; amonyum sülfat konsantrasyonu, enzim: t-butanol oranı ve pH etkisi çalışıldı. TPP sisteminin optimum saflaştırma koşulları %50(w/v) amonyum sülfat saturasyonu, 1.0:0.75 (v/v) ham ekstrakt:t-butanol oranı ve pH 4.5 olarak belirlendi. Optimize koşullar altında α-glukozidaz %60 aktivite verimi ile 3.8 kat saflaştırıldı. α-Glukozidazın molekül kütlesi sodyum dodesil sülfat poliakrilamid jel elektroforezi (SDS-PAGE) ile yaklaşık 29 kDa olarak belirlendi. Karakterizasyon çalışmaları, enzimin optimum pH ve sıcaklığının pH 4.5 ve 55°C olduğunu gösterdi. Saflaştırılan enzimin 25-60°C sıcaklık aralığında ve pH 3.5-5.5 aralığında oldukça kararlı olduğu bulundu. Kinetik sabitler (KM ve Vmax) Lineweaver-Burk diyagramından 0.64 mM ve 0.45 U olarak hesaplandı. α-Glukozidaz aktivitesi 10 mM CaCl2 konsantrasyonunda oldukça arttı (%130). TPP α-glukozidazın uygulamaları için konsantre edilmesi ve saflaştırılması için kullanışlı bir stratejidir.
Download Article in PDF (298.7 kB)