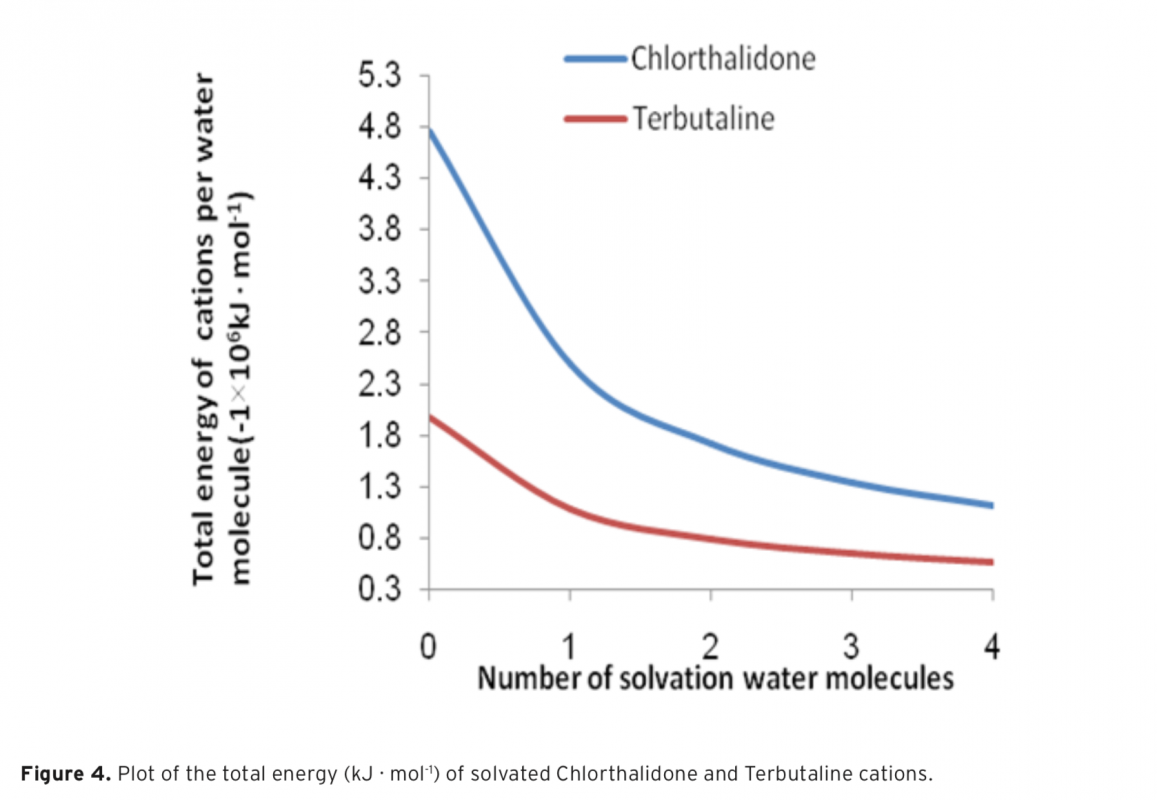
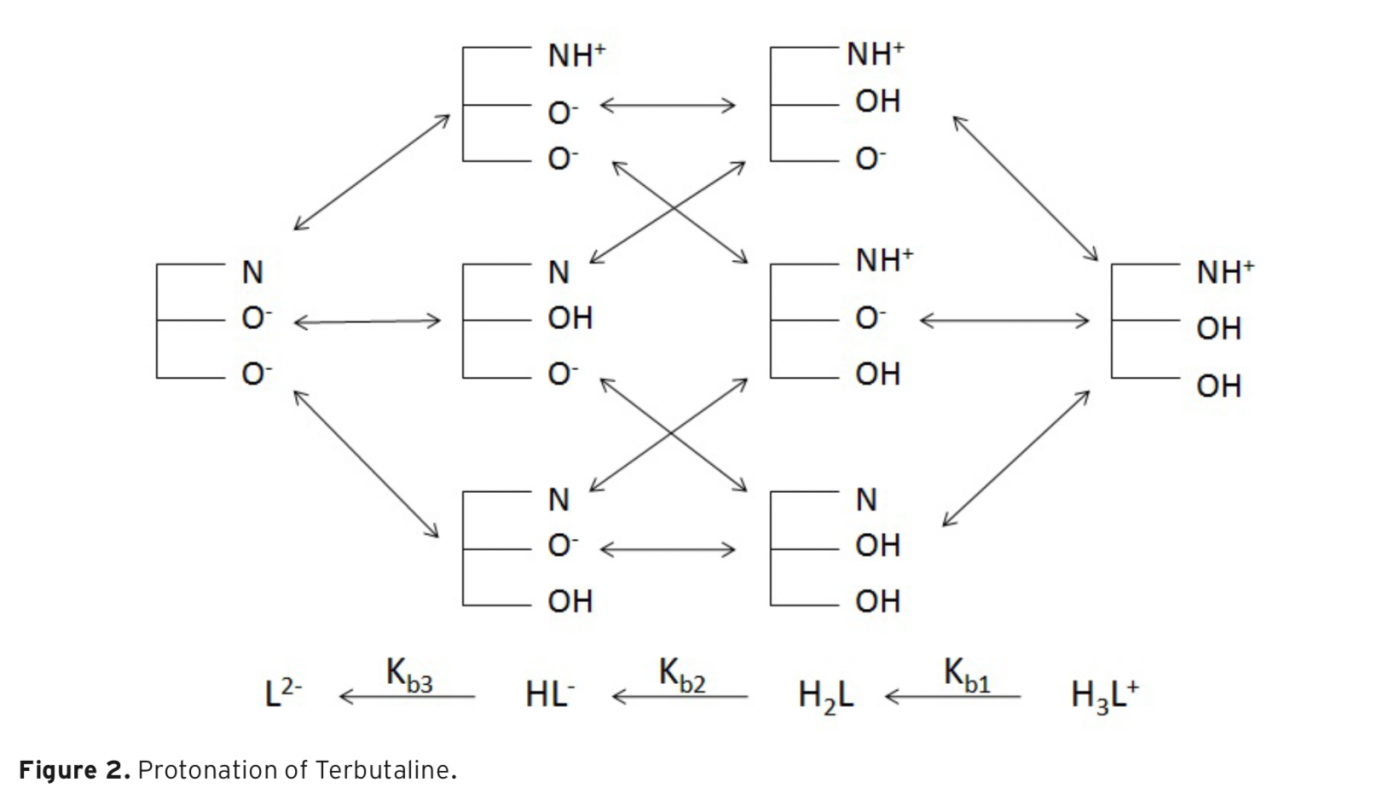
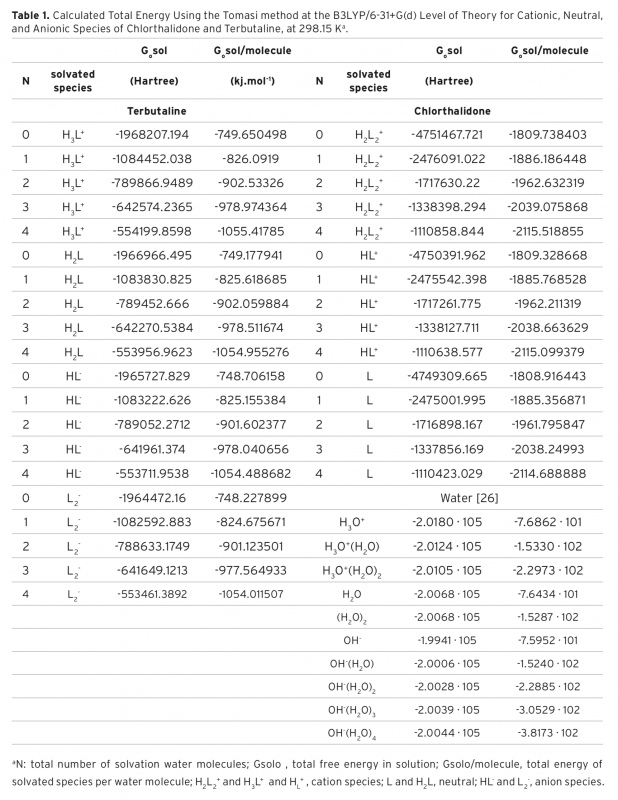
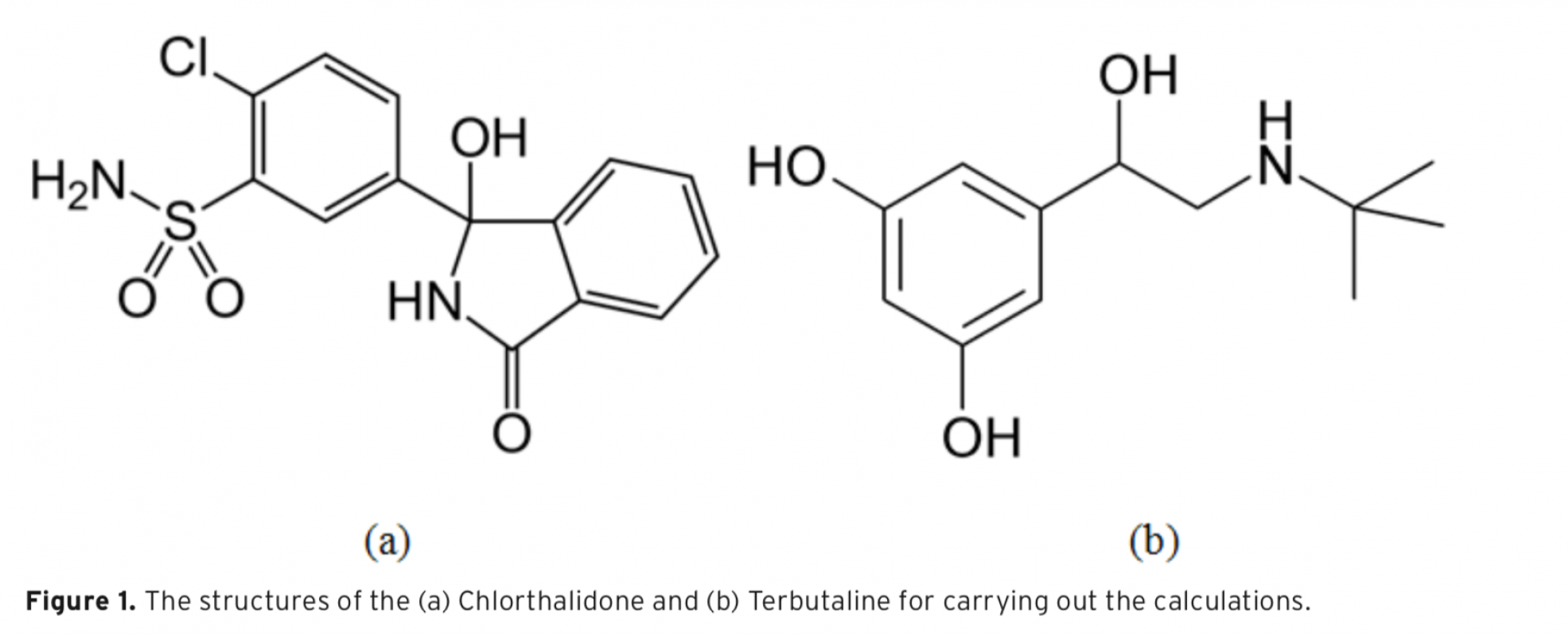
In the present study, pKa values of both drug called Chlorthalidone and Terbutaline were determined in aqueous solution. For this purpose, the B3LYP calculation with the 6-31+G(d) basis set was utilized. The reactions and equilibria that possess a high hydrogen-band-donor capability and constitute the indispensable theoretical basis to calculate the acidity constants of Chlorthalidone and Terbutaline, are shown. To analyze the formation of intermolecular hydrogen bonds between the existent species and water molecules, Tomasi’s method was used. In this way, it was determined that in alkaline aqueous solutions the cation, anion, and neutral species of Chlorthalidone and Terbutaline are solvated with one, two, three, and four molecules of water, respectively. To proceed, the calculated pKa were compared with the experimental values, which there is comparable agreement between them. The resulting data illustrated that the method was likely to be useful for the prediction of pKa values in aqueous solution.
Bu çalışmada klortalidon ve terbütalin ilaçlarının pKa değerleri sulu çözeltileri içinde hesaplanmıştır. B3LYP hesaplamaları ile 6-31+G(d) temel setinden faydalanmak amaçlanmıştır. Hidrojen bağı verici özelliği yüksek olan ve aynı zamanda klortalidon ve terbütalin için asitlik sabitlerinin hesaplanmasında teorik açıdan vazgeçilmez olan denge ve tepkimeler gösterilmiştir. Var olan türler ve su molekülleri arasındaki hidrojen bağı oluşumlarını analiz etmek için Tomasi yöntemi kullanılmıştır. Böylece klortalidon ve terbütalinin alkali su çözeltisi içerisindeki katyon, anyon ve nötr oluşumlarının, sırasıyla bir, iki, üç ve dört su molekülüyle çözünebildiği saptanmıştır. Sonraki adımda hesaplanan pKa değerleri deneysel sonuçlarla karşılaştırılmış ve kabul edilebilir bir örtüşme görülmüştür. Elde edilen veriler uygulanan tekniğin sulu çözeltilerde pKa tahminlerinde kullanışlı bir seçenek olacağını göstermektedir.
Download Article in PDF (875.5 kB)