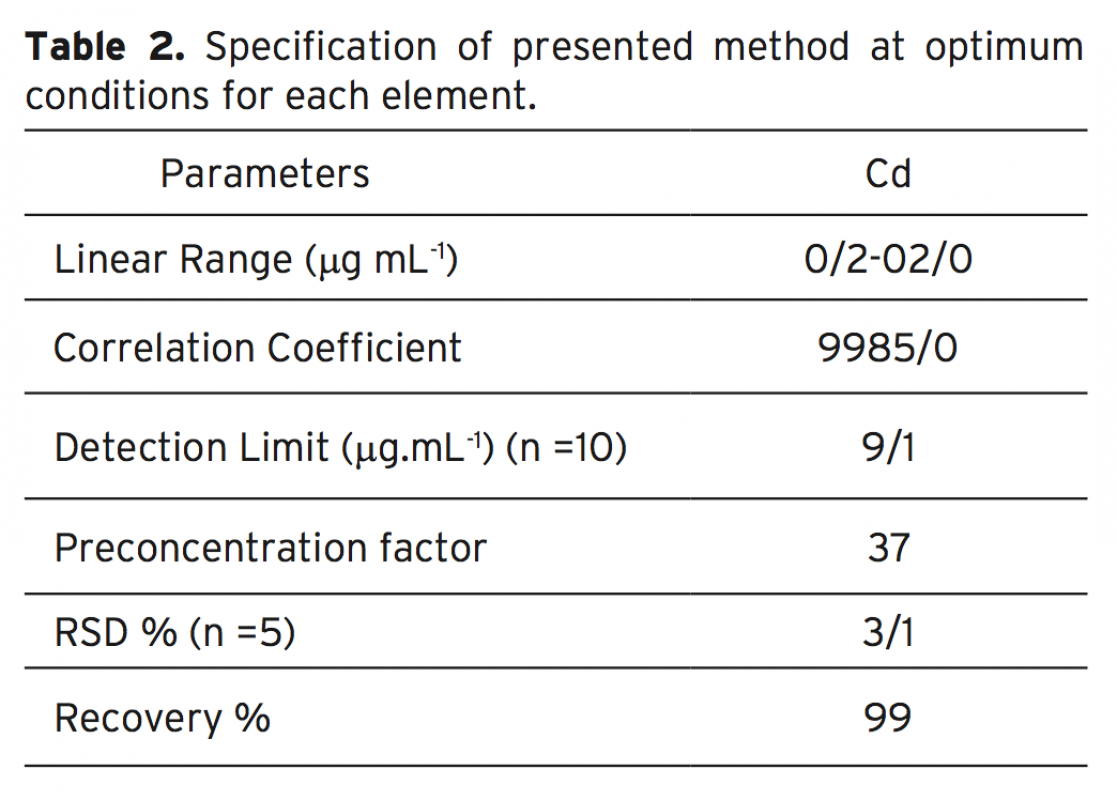
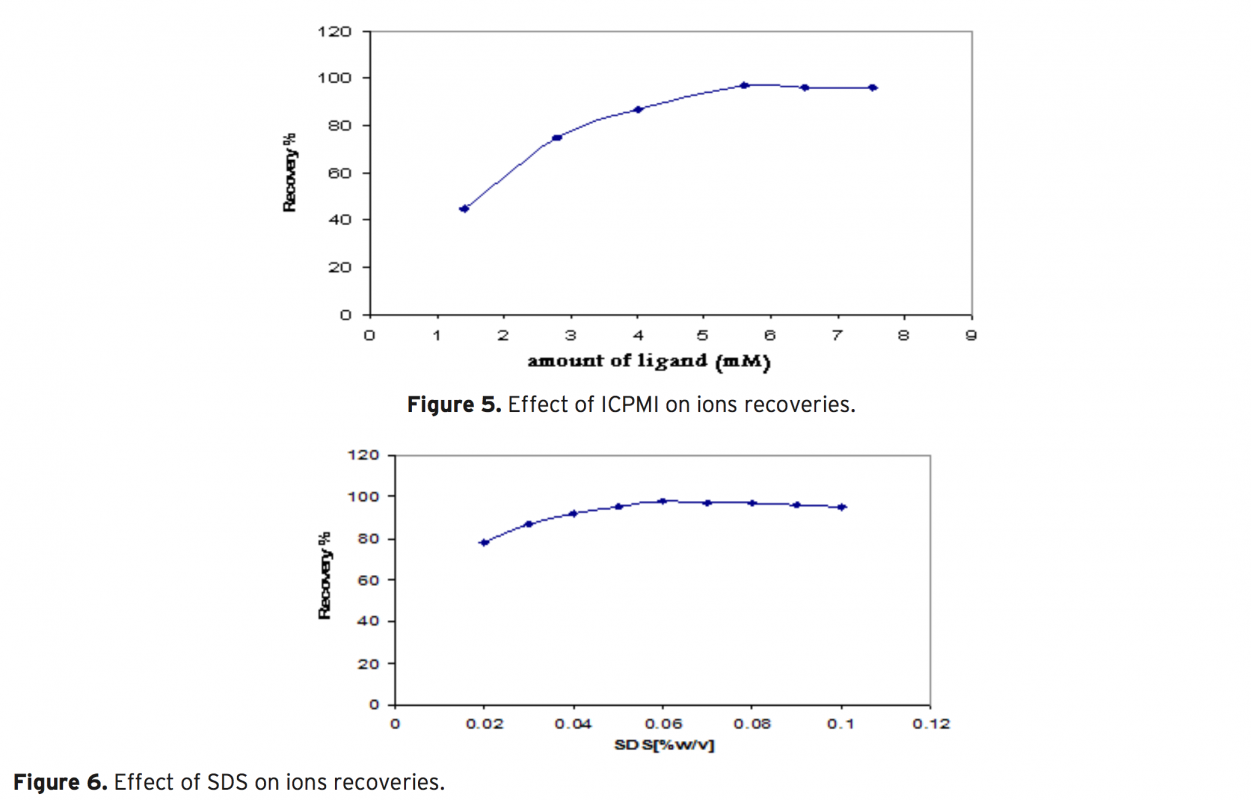
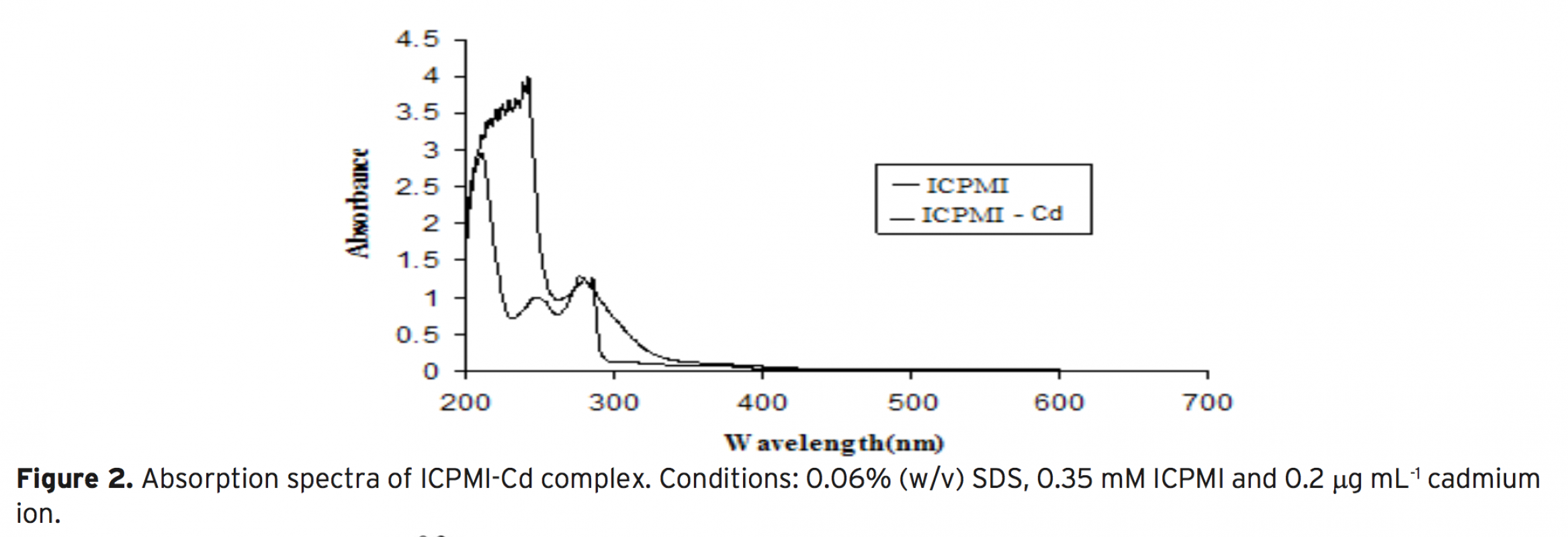
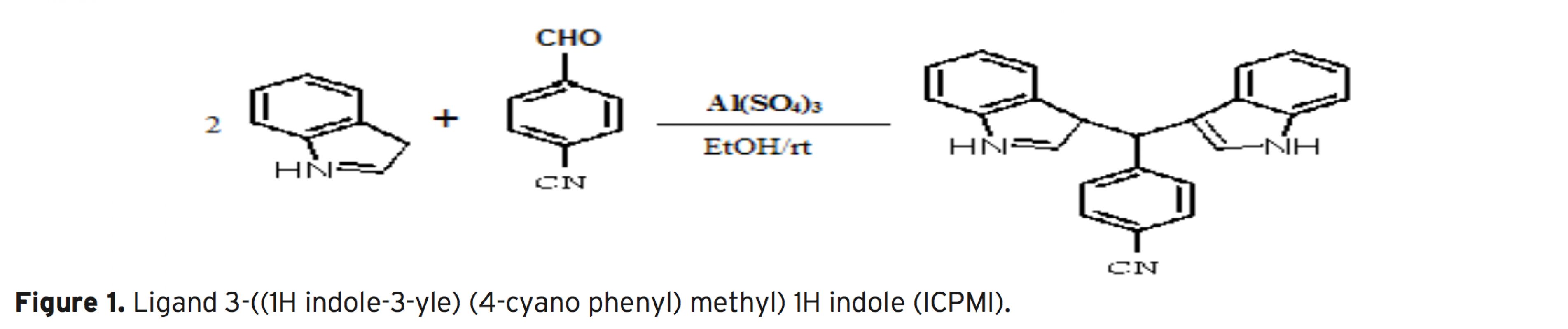
An efficient flotation method based on the combination of flame atomic absorption spectrometry (FAAS) and separation and preconcentration step for determination of Cd2+ ions in various real samples by the possibility of applying Ligand 3-((1H indole-3-yle) (4-cyano phenyl) methyl) 1H indole (ICPMI), as a new collec- tor was studied. The influence of pH, amount of ICPMI as collector, sample matrix, type and amount of eluting agent, type and amount of surfactant as floating agent, ionic strength and air flow rates i.e. variables affecting the efficiency of the extraction system was evaluated. It is as certained that cadmium ion separated simulta- neously from matrix in the presence of 0.012 mM ligand, 0.025% (w/v) of sodium dodecyl sulfate (SDS) and pH 9.0±0.1. These ions can be eluted quantitatively with 0.5 mL of 1M HNO3 in methanol linear range of the determination is between 0.02-2.2 μgmL-1 for Cd2+ with a detection limit for Cd2+ was1.9 μg mL-1 respectively. The method has been successfully applied for determination of trace amounts of cadmium ion in in radiology wastewater, amalgam, natural water and blood samples samples.
Alev atomik absorpsiyon spektrometrisine dayanan etkin bir flotasyon yöntemiyle ayrım ve ön deriştirme adımları toplayıcı olarak ligand 3-((1H-indol-3-il) (4-Siyano fenil) metil) lH-indol (ICPMI) ile çeşitli gerçek örneklerde Cd2+ iyonlarının tayini incelenmiştir. pH, ICPMI miktarı, numune matrisi, elüsyon maddesinin türü ve miktarı, yüzey aktif maddenin türü ve miktarı, iyonik kuvvet, hava akış oranı, yani ekstraksiyon sisteminin verimliliğini etkileyen değişkenler değerlendirildi. Kadmiyum iyonunun 0.012 mM ligandı, %0.025 (w/v) sodyum dodesil sülfat (SDS) ve pH 9.0±0.1 varlığında eşzamanlı olarak matristen ayrıldığı kesin olarak belirlendi. Bu iyonlar, metanolde 1M HNO3’ün 0.5 mL si ile nicel olarak, Cd2+ için 0.02-2.2 μgmL-1 arasında ve Cd2+ için saptama limiti 1.9 μgmL-1 olan doğrusal aralıklarla ayrılabilir. Yöntem, radyoloji atık suyu, amalgam, doğal su ve kan numuneleri örneklerinde az miktarda kadmiyum iyonu tayini için başarıyla uygulanmıştır.
Download Article in PDF (230.4 kB)