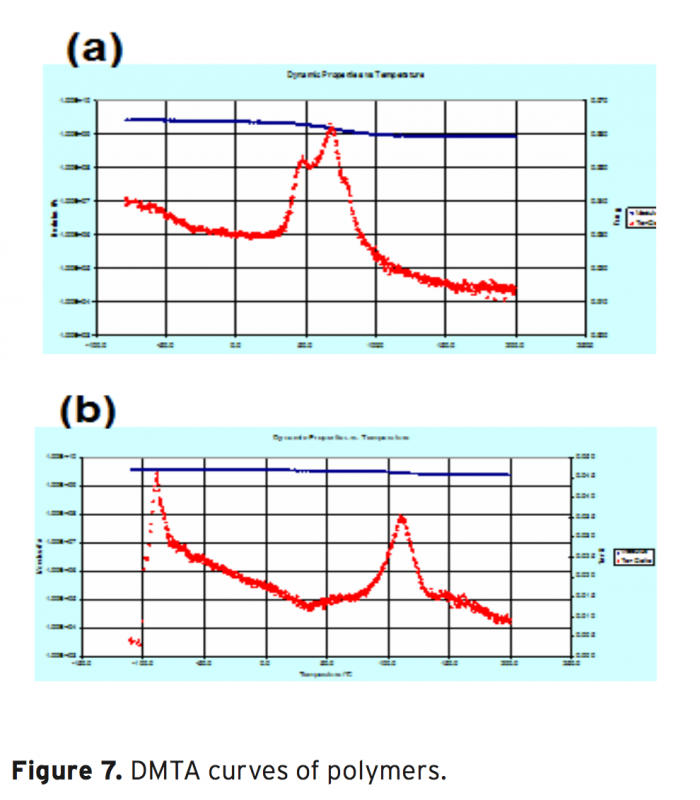
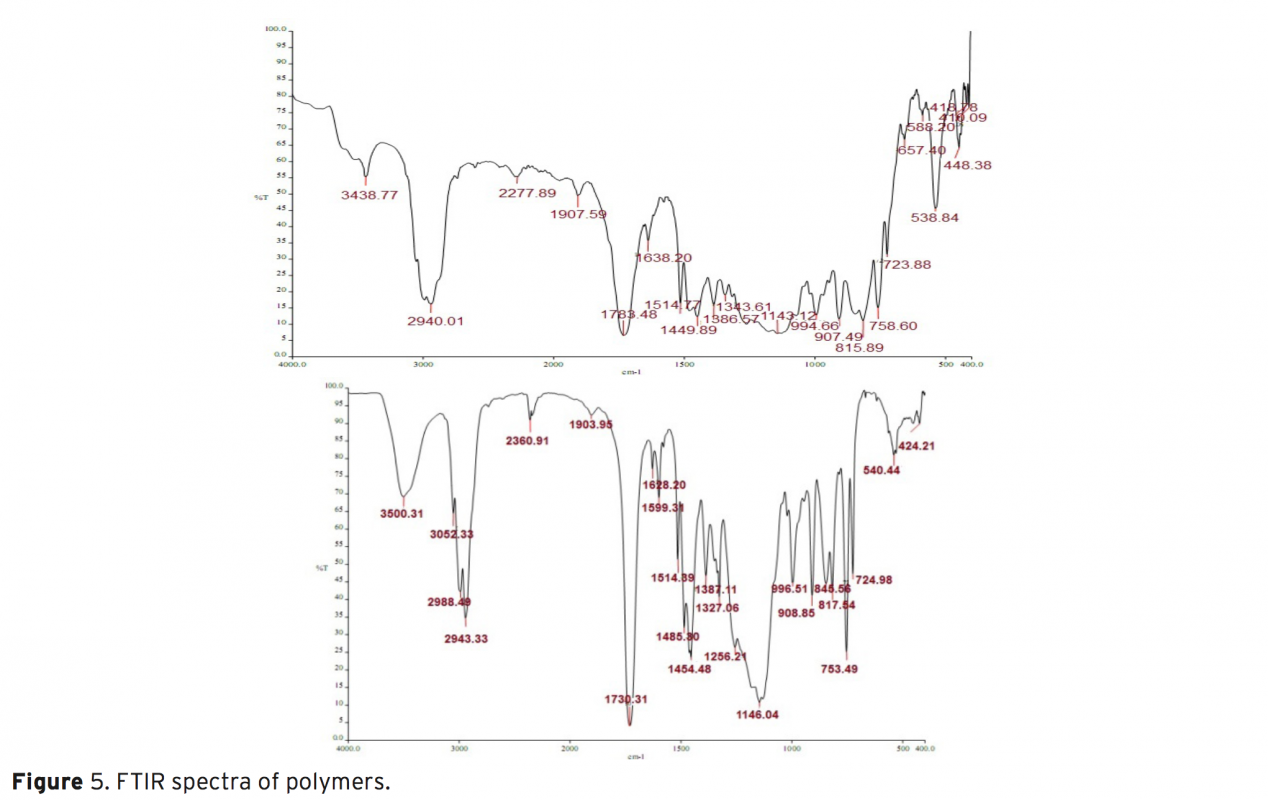
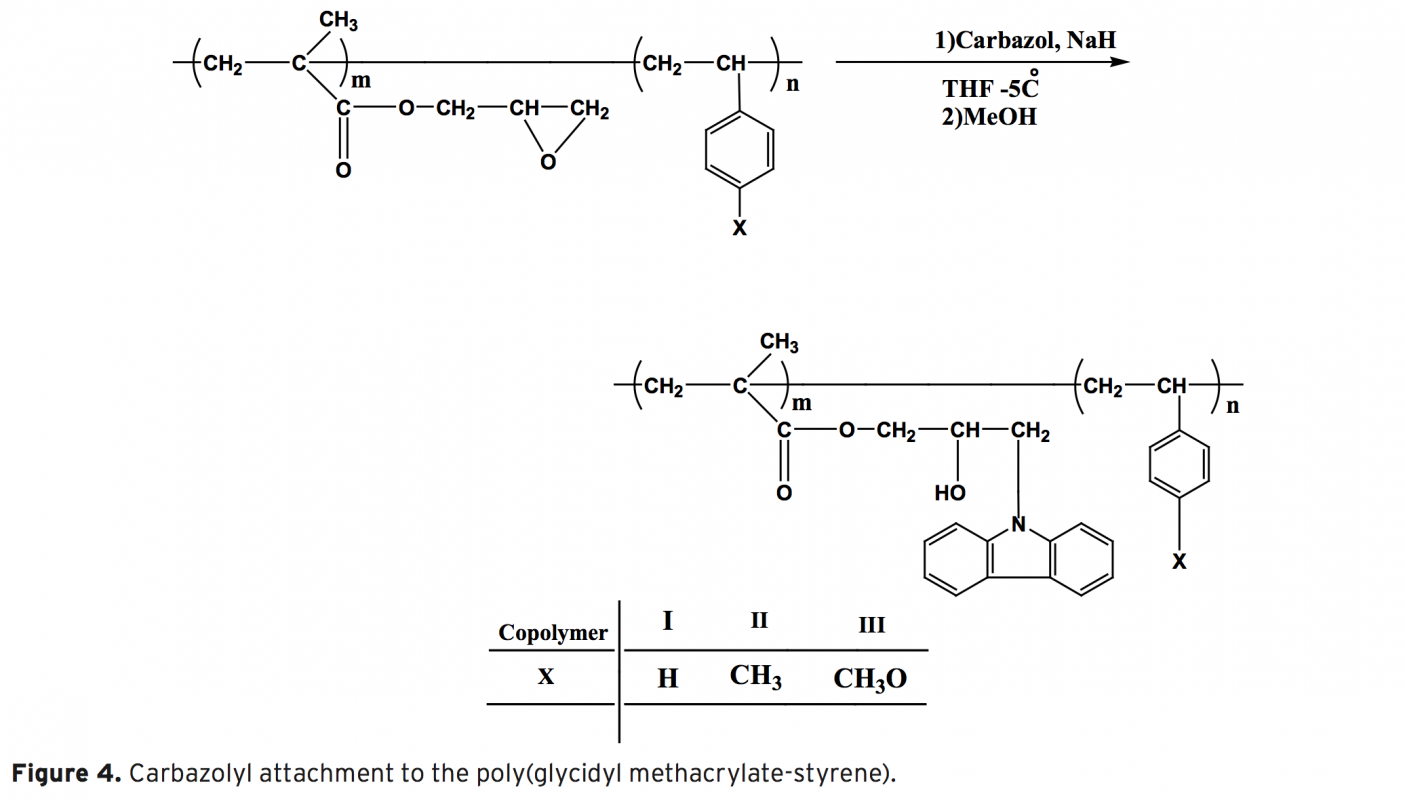
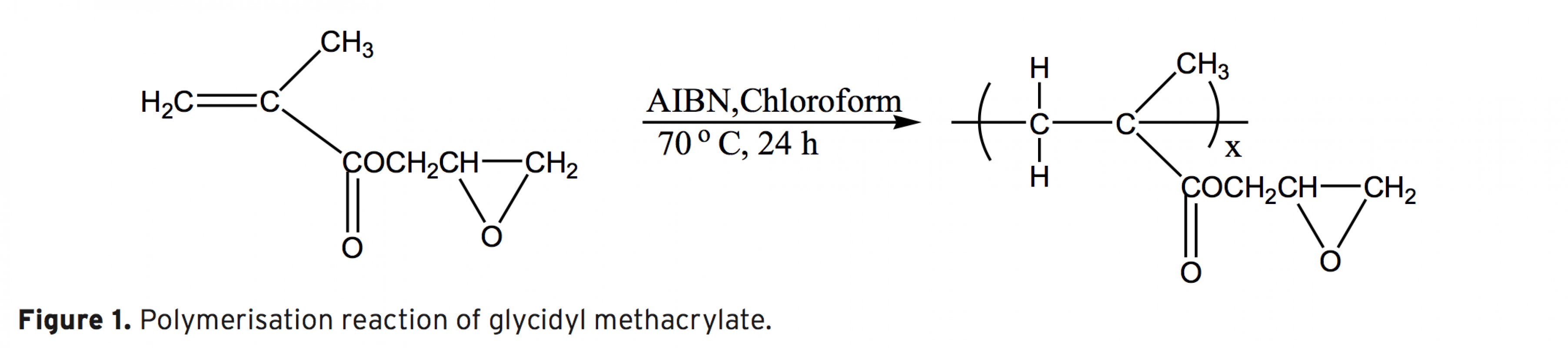
The synthesis, characterization and thermal behaviour of styrenic copolymers of glycidyl methacrylate without and with carbazole groups are reported. The copolymers based on glycidyl methacrylates belong to the potential class of functional polymers. Epoxide ring opening reaction is a route that explains the chemical modification of glycidyl methacrylate (GMA) polymers with nucleophilic reagent containing hydroxyl, carboxyl or amine groups. In this work, copolymers of GMA have been modified by incorporation of bulky carbazole groups.The homopolymer of glycidyl methacrylate (GMA) and its random copolymers with styrene , methyl styrene, methoxy styrene were synthesized by free radical polymerization using azobis(isobutyronitrile) (AIBN) as initiator at 70±1°C. The polymers were characterized by 1H-NMR and IR spectroscopic techniques. This modification increases the rigidity of polymers and, subsequently, their glass transition temperature of polymer as shown by DMTA analysis. The results show that, with the incorporation of the carbazole groups in the polymer side chains, a series of novel modified polymer containing new properties are obtained, that can find some applications in polymer industry.
Karbazol grupları varlığında ve yokluğunda glisidil metakrilatın stirenik kopolimerlerinin sentezi, karakterizasyonu ve ısıl davranışları rapor edilmiştir. Glisidil metakrilat temelli kopolimerler fonksiyonel polimerler sınıfına aittir. Epoksit halka açma tepkimesi, hidroksil, karboksil veya amin grupları içeren nükleofilik reaktif ile glisidil metakrilat (GMA) polimerlerinin kimyasal modifikasyonunu açıklayan bir yoldur. Bu çalışmada, GMA kopolimerleri büyük karbazol gruplarının birleşmesiyle modifiye edilmiştir. Glisidil metakrilat (GMA) homopolimeri ve stiren, metil stiren, metoksi stirenli rasgele kopolimerler 70±1°C’da başlatıcı olarak azobis(izobütironitril) (AIBN) kullanılarak serbest radikal polimerizasyonu ile sentezlendi. Polimerler, 1H-NMR ve IR spektroskopik teknikleriyle karakterize edildi. Bu modifikasyon, polimerlerin sertliğini ve ardından DMTA analizi ile gösterilen polimerlerin camsı geçiş sıcaklığını arttırır. Sonuçlar, karbazol gruplarının polimer yan zincirlerine dahil edilmesi ile, polimer endüstrisinde bazı uygulamalar bulabilen yeni özelliklere sahip bir dizi yeni modifiye edilmiş polimer elde edildiğini göstermektedir.
Download Article in PDF (414.8 kB)