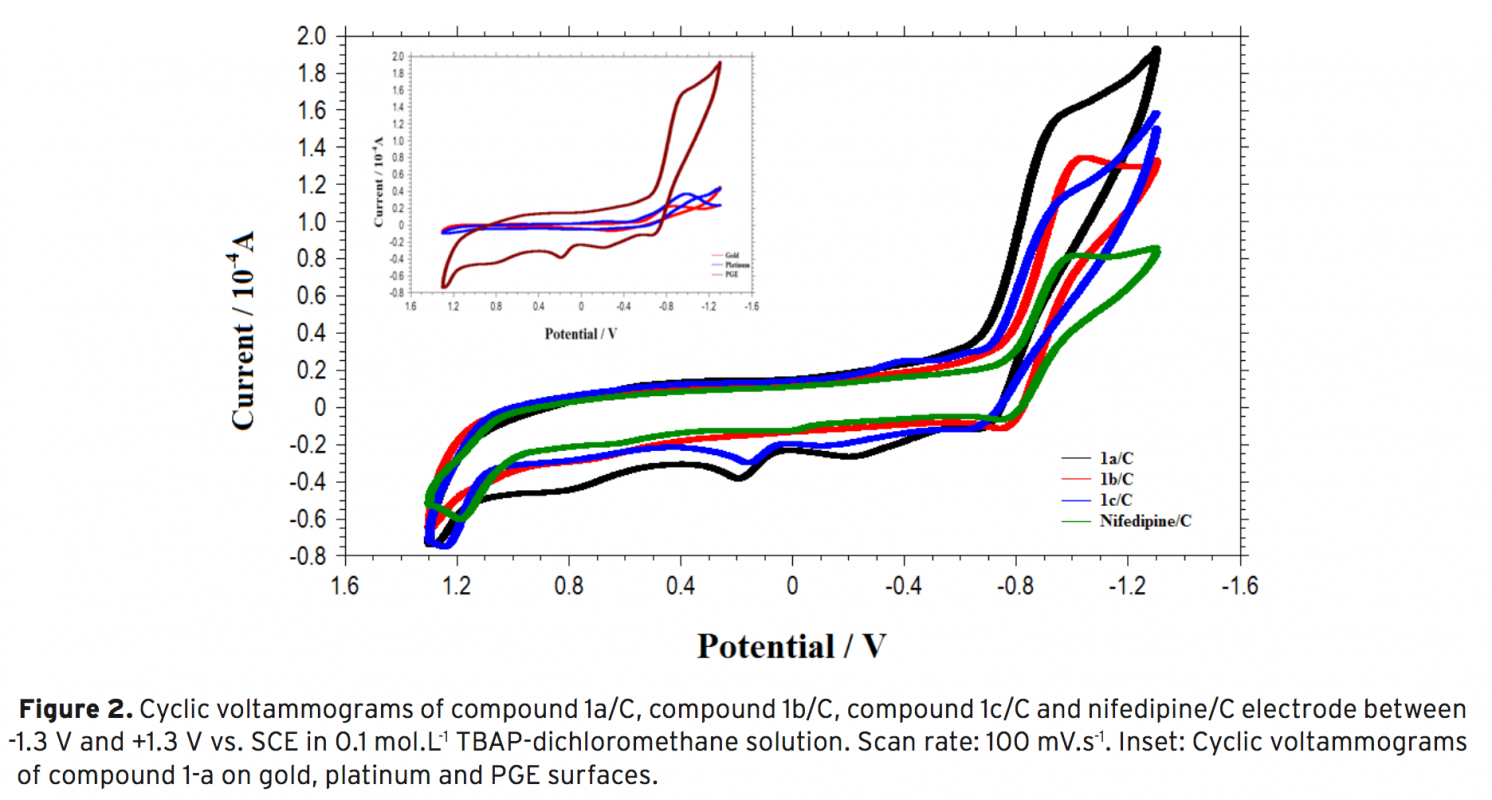
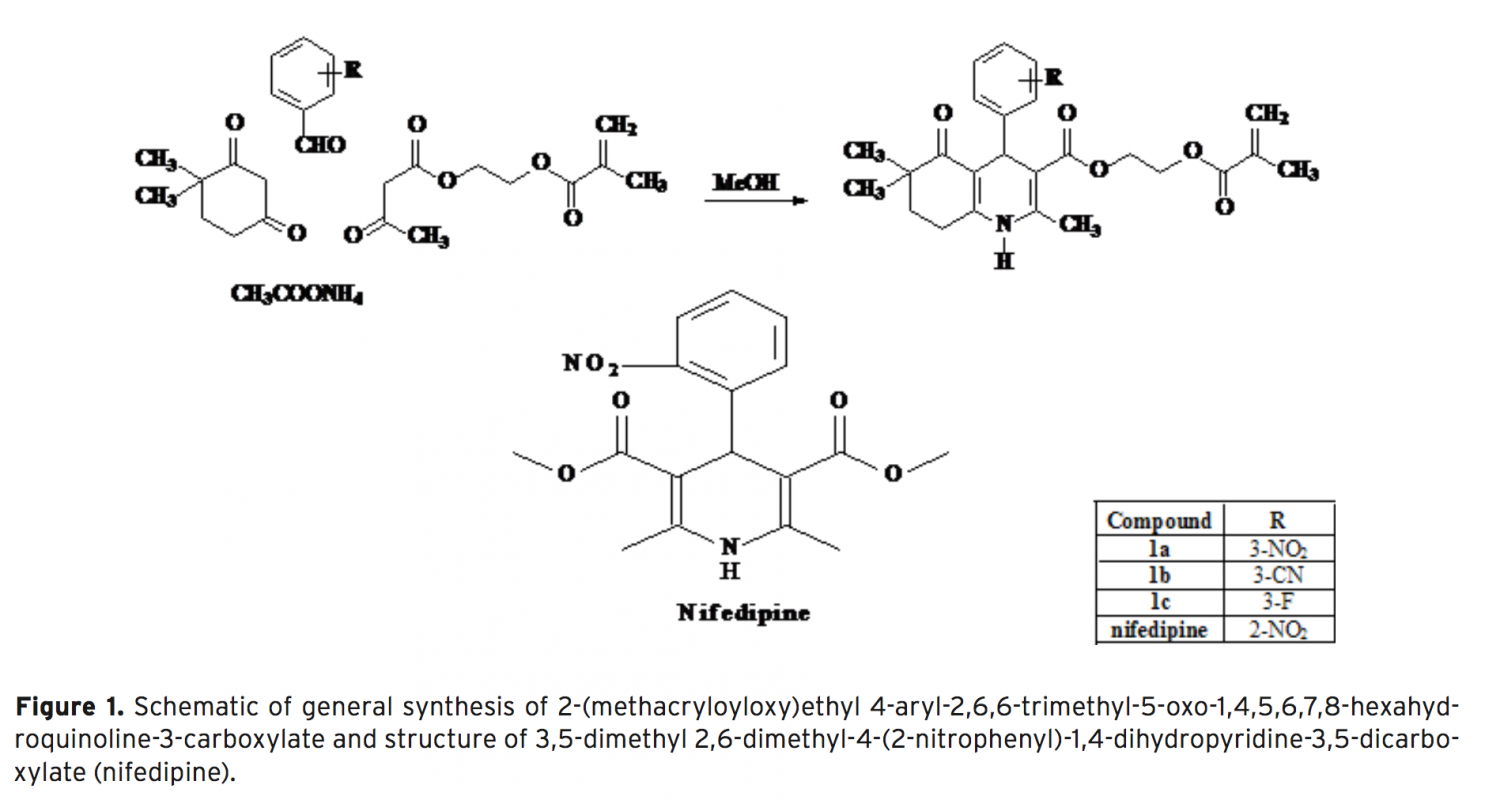
Antioxidant is a molecule which retards the autooxidation of fats and oils. Antioxidants block the oxidation process which can cause damage to many cells in body helping to prevent diseases. Therefore, the importance of antioxidant is in increasing demand. So far many attempts have been carried out for developing to detect antioxidant activities. Among various analytical techniques, electrochemistry has been a more approp- riate way to determine antioxidant activity due to its ease of use and short-lasting application. In this study, it was aimed to determine antioxidant activities of recently synthesized condensed 1,4-dihydropyridine derivatives using disposable pencil graphite electrode. Cyclic voltammetry technique was used to determine oxidation potential value, and also differential pulse voltammetry technique was used to determine reduction potential values of the derivatives. To verify our results, nifedipine was used as a reference drug and conventional antioxidant activities were also used as a basis for comparison.
Antioksidan katı ve sıvı yağların otoksidasyonunu geciktiren bir moleküldür. Antioksidanlar, hastalıkları engellemeye yardımcı olarak vücutta pekçok hücreye zarar verebilen oksidasyon sürecini bloke ederler. Bu yüzden, antioksidanın önemine olan talep giderek artmaktadır. Bugüne kadar, pek çok teşebbüs antioksidan aktivite tayininin geliştirilmesi için gerçekleştirilmiştir. Çok çeşitli analitik tekniklerin arasında, elektrokimya kullanım kolaylığına ve kısa sürede uygulanabilmesine bağlı olarak antioksidan aktivitesinin belirlenmesi için daha uygun bir yoldur. Bu çalışmada, yeni sentezlenen kondanse 1,4-dihidropiridin türevlerinin antioksidan aktivitelerinin atılabilir kalem grafit elektrot kullanılarak belirlenmesi amaçlanmıştır. Dönüşümlü voltametri tekniği türevlerin oksidasyon potansiyel değerinin ve aynı zamanda diferansiyel puls voltametri tekniği redüksiyon potansiyel değerinin belirlenmesi için kullanılmıştır. Sonuçlarımızın değerlendirilmesi için, nifedipin bir referans ilaç olarak ve konvensiyonel antioksidan aktiviteleri aynı zamanda karşılaştırma için bir temel olarak kullanılmıştır.
Download Article in PDF (472.4 kB)