Waste water treatment systems have great importance as a result of the increasing population and environ- mental pollution. In this study, electrosorptive removal of benzoic acid (BA) and nicotinic acid (NA) from aqueous solutions onto activated carbon electrodes (ACEs) were studied. ACEs were fabricated from the in-
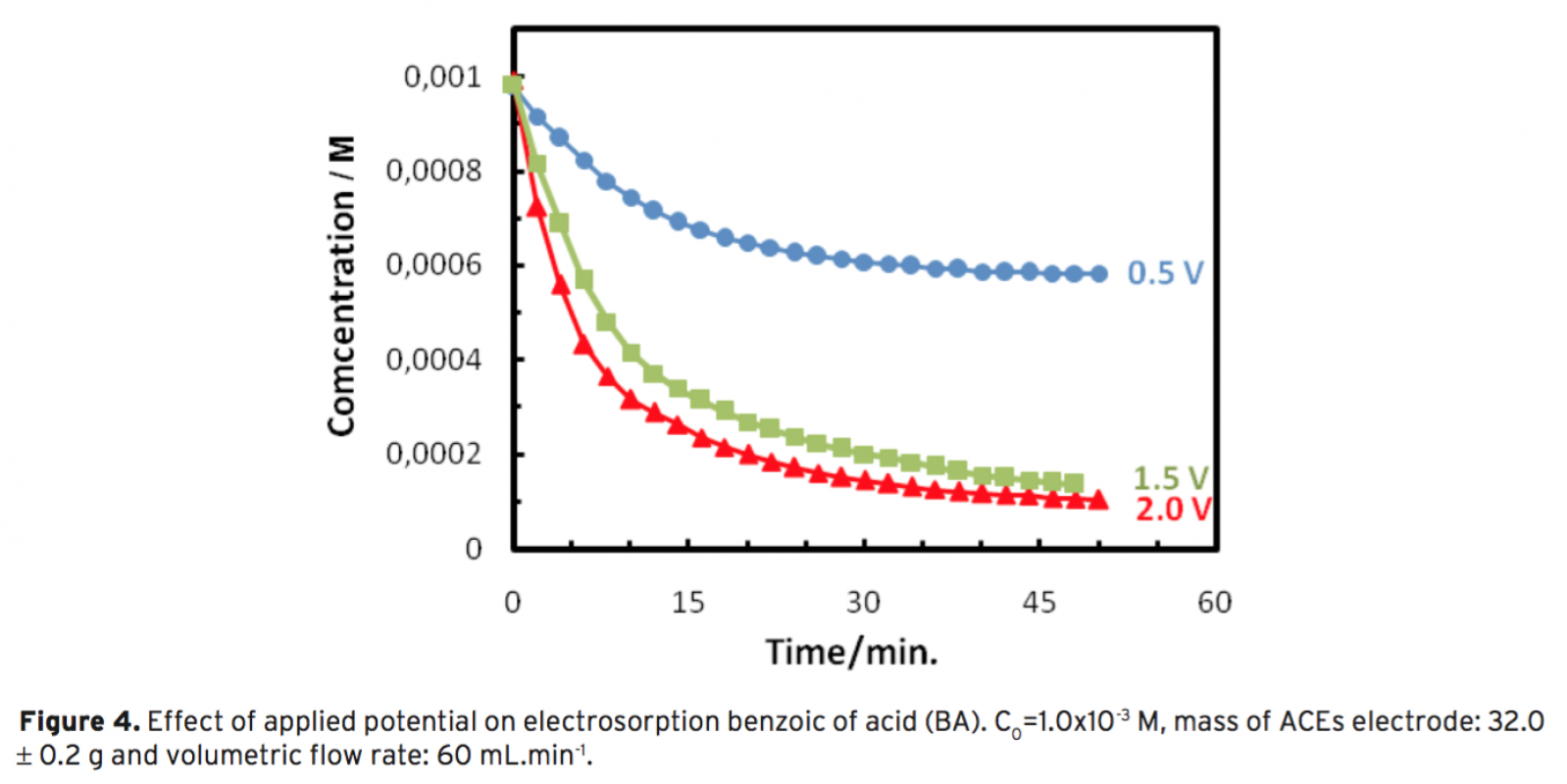
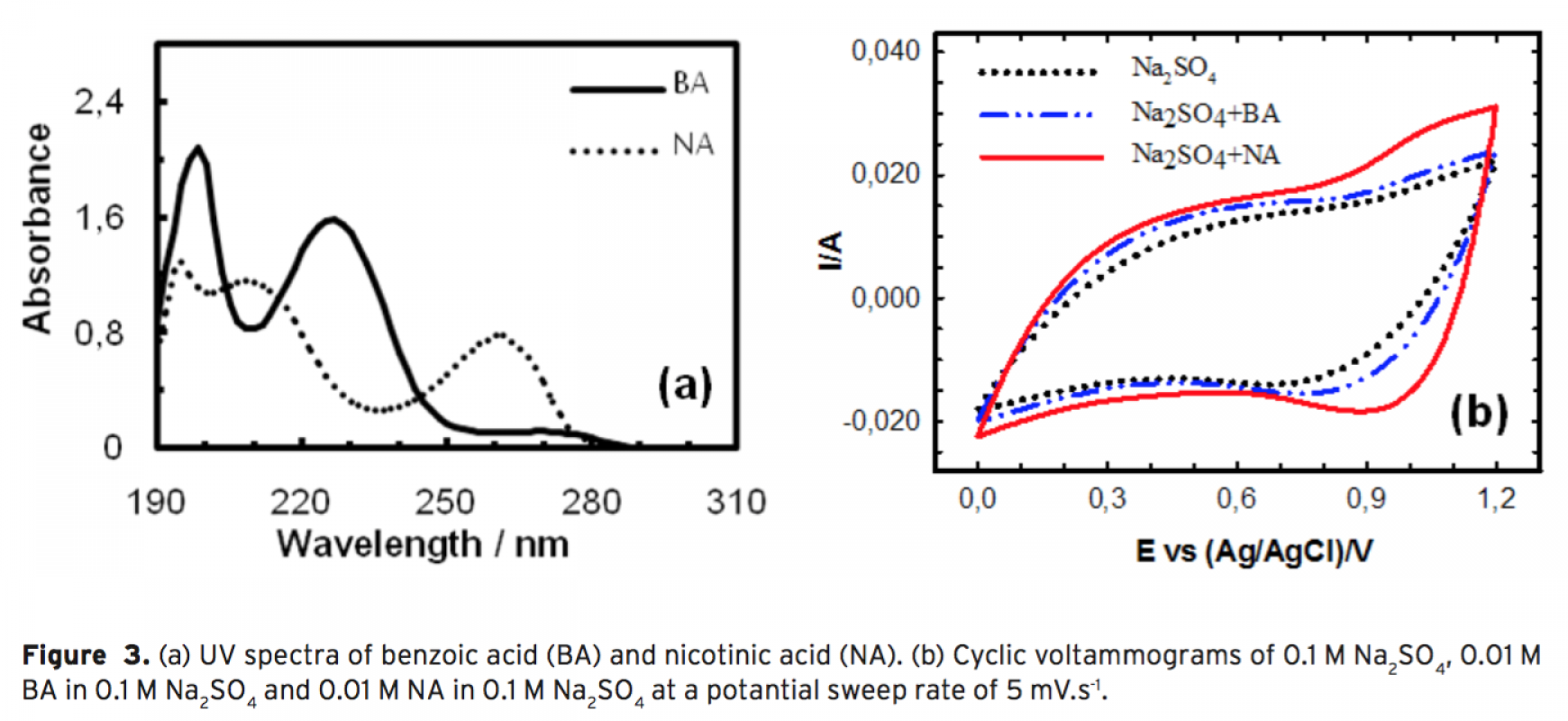
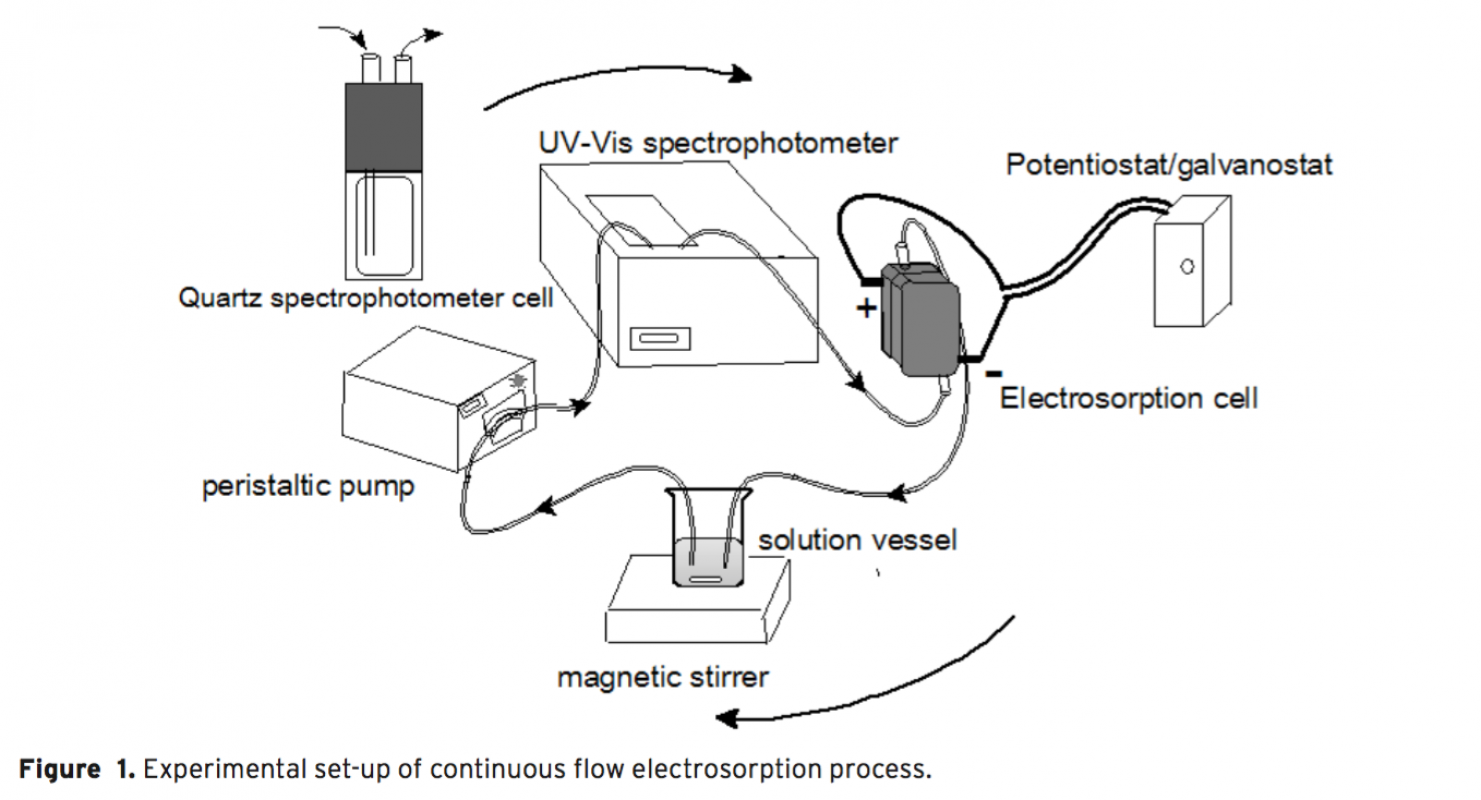
dustrially available granular activated carbon and polyethylene as binder. During the electrosorption process, the change in concentration of organic molecules was followed by using an online UV–Vis spectrophotometric system. Effects of operational parameters such as applied potential and volumetric flow rate on electrosorp- tion of BA were examined. Optimum parameters for the process were found to be 60 mL.min-1 for volumetric flow rate and 1.5 V for polarization of ACEs. Electrosorption of binary mixtures of BA and NA was also studied. it can be concluded that BA and NA can be effectively removed by electrosorption from aqueous solutions using activated carbon electrodes.
Su arıtma sistemleri, artan insan nüfusu ve çevre kirliliği sonucunda oldukça önemli hale gelmiştir. Bu çalışmada, benzoik asit (BA) ve nikotinik asitin (NA) sulu çözeltilerinden, aktiflenmiş karbon elektrotlar (ACEs) üzerine elektrosorpsiyon yolu ile uzaklaştırılmaları incelenmiştir. ACEs, endüstriyel olarak elde edilebilen
granüllü aktiflenmiş karbon ve polietilen bağlayıcı kullanılarak üretilmiştir. Elektrosorpsiyon sırasında organik moleküllerin derişimlerinde oluşan değişimler, online UV–Vis spektrofotometrik sistem kullanılarak takip edilmiştir. BA nın elektrosorsiyonu üzerine uygulanan potansiyel ve çözelti akış hızı gibi sistem parametrelerinin etkileri incelenmiştir. Optimum proses parametrelerinin, çözelti akış hızı için 60 mL.dk-1 ve ACEs polarizasyonu için 1.5 V olduğu bulunmuştur. BA ve NA içeren ikili karışımların elektrosorpsiyon davranışları da incelenmiştir. BA ve NA nın aktiflenmiş karbon elektrotlar kullanılarak sulu çözeltilerinden elektrosorpsiyon ile etkili bir şekilde uzaklaştırılabileceği sonucuna varılmıştır.

Download Article in PDF (307.4 kB)